Please select a subject first
Advertisements
Advertisements
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
Concept: undefined >> undefined
What are micelles? Give an example of a micellers system.
Concept: undefined >> undefined
Advertisements
Calculate the overall complex dissociation equilibrium constant for the Cu(NH3)42+ ion, given that β4 for this complex is 2.1 × 1013.
Concept: undefined >> undefined
What is meant by stability of a coordination compound in solution?
Concept: undefined >> undefined
Where does the water present in the egg go after boiling the egg?
Concept: undefined >> undefined
What is the effect of denaturation on the structure of proteins?
Concept: undefined >> undefined
Write a short note on diazotisation.
Concept: undefined >> undefined
Give the IUPAC name of the following compound:
PhCH2CH2COOH
Concept: undefined >> undefined
Give the IUPAC name of the following compound:
(CH3)2C=CHCOOH
Concept: undefined >> undefined
Give the IUPAC name of the following compound:

Concept: undefined >> undefined
Give the IUPAC name of the following compound:
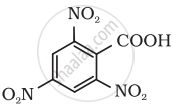
Concept: undefined >> undefined
What type of colloid is formed when a gas is dispersed in a liquid? Give an example
Concept: undefined >> undefined
How do you convert toluene to benzoic acid?
Concept: undefined >> undefined
Define the following with an example
Denatured protein
Concept: undefined >> undefined
Give reasons Dioxygen is a gas while Sulphur is a solid at room temperature.
Concept: undefined >> undefined
How do you convert the following :
Ethyne to Ethanal
Concept: undefined >> undefined
How do you convert the following:
Acetic acid to Methane
Concept: undefined >> undefined
Write the equations involved in the following reactions :
Stephen reaction
Concept: undefined >> undefined
Write the equations involved in the following reactions :
Wolff-Kishner reduction
Concept: undefined >> undefined
Calculate the mass of NaCl (molar mass = 58.5 g mol−1) to be dissolved in 37.2 g of water to lower the freezing point by 2°C, assuming that NaCl undergoes complete dissociation. (Kf for water = 1.86 K kg mol−1)
Concept: undefined >> undefined
