Advertisements
Advertisements
प्रश्न
How do you convert the following:
Acetic acid to Methane
How will you convert the following: (Give a balanced equation)
Acetic acid to methane.
Advertisements
उत्तर
Acetic acid can be converted to methane in the following steps:
Step 1: Ethanoic acid is treated with NaOH to obtain sodium ethanoate salt and water.
\[\ce{\underset{Ethanoic acid}{CH3COOH} + NaOH -> \underset{Sodium ethanoate}{CH3COONa} + H2O}\]
Step 2: Sodium ethanoate is heated strongly with soda lime (NaOH + CaO) to obtain methane and S.
\[\ce{CH3COONa_{(s)} + NaOH_{(s)} -> \underset{Methane}{CH4} + \underset{Sodium carbonate}{Na2CO3}}\]
संबंधित प्रश्न
Write the structures and IUPAC names of the :Adipic acid compound
Give the IUPAC name of the following compound:
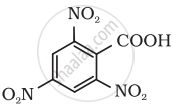
Complete and rewrite the balanced chemical equations
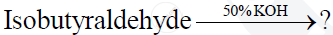
How do you convert toluene to benzoic acid?
Identify the compounds A, B, C and D.
How do you convert the following :
Ethyne to Ethanal
Write the equations involved in the following reactions :
Stephen reaction
Write the equations involved in the following reactions :
Wolff-Kishner reduction
Complete the following:
\[\ce{C6H5NO2 ->[Sn/HCl] A ->[Br2/H2O] B ->[NaNO2/HCl][273 - 278 K] C ->[HBF4][Δ] D}\]
The correct order for the acidic character of the following carboxylic acids is:
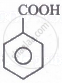 |
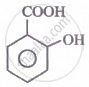 |
 |
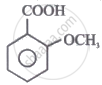 |
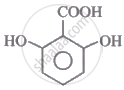 |
| (I) | (II) | (III) | (IV) | (V) |
