Please select a subject first
Advertisements
Advertisements
What is the role of depressants in the froth floatation process?
Concept: undefined >> undefined
Complete the following equations:
`2MnO_4^(-)+16H^++5S^(2-)rarr`
Concept: undefined >> undefined
Advertisements
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Concept: undefined >> undefined
Write the fomulae of the following compounds :
Tetraaquodichlorochromium (III) chloride
Concept: undefined >> undefined
Write the fomulae of the following compounds
Potassium tetracyanoaurate (III) ion
Concept: undefined >> undefined
Write the product when D-glucose reacts with conc. HNO3.
Concept: undefined >> undefined
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Concept: undefined >> undefined
Name the reagents used in the following reactions:

Concept: undefined >> undefined
Complete the following equation :
Concept: undefined >> undefined
Write the structures of A, B and C in the following reactions :
Concept: undefined >> undefined
A first order reaction takes 10 minutes for 25% decomposition. Calculate t1/2 for the reaction.
(Given : log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
Concept: undefined >> undefined
Show that the time required for 99% completion is double of the time required for the completion of 90% reaction.
Concept: undefined >> undefined
Which of the following isomers is more volatile:
o-nitrophenol or p-nitrophenol?
Concept: undefined >> undefined
Complete the following chemical equations :
Cu + H2SO4(conc.) →
Concept: undefined >> undefined
In reference to Freundlich adsorption isotherm, write the expression for adsorption of gases on solids in the form of an equation.
Concept: undefined >> undefined
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
Concept: undefined >> undefined
Give the structures of A, B and C in the following reactions :
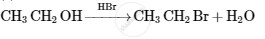
Concept: undefined >> undefined
Calculate emf of the following cell at 298 K:
\[\ce{Mg_{(s)} | Mg^{2+} (0.1 M) || Cu^{2+} (0.01) | Cu_{(s)}}\]
[Given \[\ce{E^{\circ}_{cell}}\] = +2.71 V, 1 F = 96500 C mol–1]
Concept: undefined >> undefined
Name a member of the lanthanoid series that is well-known to exhibit +2 oxidation state.
Concept: undefined >> undefined
Give the structures of A, B and C in the following reactions :

Concept: undefined >> undefined
