Please select a subject first
Advertisements
Advertisements
Match the pollutants given in Column I with their effects given in Column II.
| Column I | Column II |
| (i) Phosphate fertilisers in water | (a) BOD level of water increases |
| (ii) Methane in air | (b) Acid rain |
| (iii) Synthetic detergents in water | (c) Global warming |
| (iv) Nitrogen oxides in air | (d) Eutrophication |
Concept: undefined >> undefined
Assertion (A): Green house effect was observed in houses used to grow plants and these are made of green glass.
Reason (R): Green house name has been given because glass houses are made of green glass.
Concept: undefined >> undefined
Advertisements
Assertion (A): The pH of acid rain is less than 5.6.
Reason (R): Carbon dioxide present in the atmosphere dissolves in rain water and forms carbonic acid.
Concept: undefined >> undefined
Assertion (A): Carbon dioxide is one of the important greenhouse gases.
Reason (R): It is largely produced by respiratory function of animals and plants.
Concept: undefined >> undefined
Green plants use carbon dioxide for photosynthesis and return oxygen to the atmosphere, even then carbon dioxide is considered to be responsible for greenhouse effect. Explain why?
Concept: undefined >> undefined
Explain how does greenhouse effect cause global warming.
Concept: undefined >> undefined
Which of the following is a secondary pollutant?
Concept: undefined >> undefined
What is the basic difference in approach between the Mendeleev’s Periodic Law and the Modern Periodic Law?
Concept: undefined >> undefined
On the basis of quantum numbers, justify that the sixth period of the periodic table should have 32 elements.
Concept: undefined >> undefined
In the modern periodic table, the period indicates the value of:-
Concept: undefined >> undefined
Which of the following statements related to the modern periodic table is incorrect?
Concept: undefined >> undefined
The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
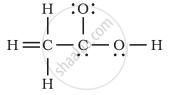
Concept: undefined >> undefined
In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process?
Concept: undefined >> undefined
How is classical smog different from photochemical smog?
Concept: undefined >> undefined
Write down the reactions involved during the formation of photochemical smog.
Concept: undefined >> undefined
What are the harmful effects of photochemical smog?
Concept: undefined >> undefined
What are electron deficient compounds? Are BCl3 and SiCl4 electron deficient species? Explain
Concept: undefined >> undefined
In what ways lithium shows similarities to magnesium in its chemical behaviour?
Concept: undefined >> undefined
Why is Li2CO3 decomposed at a lower temperature whereas Na2CO3 at a higher temperature?
Concept: undefined >> undefined
