Please select a subject first
Advertisements
Advertisements
Account for the following observations:
PbO2 is a stronger oxidising agent than SnO2
Concept: undefined >> undefined
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Concept: undefined >> undefined
Advertisements
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
Concept: undefined >> undefined
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Concept: undefined >> undefined
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
Concept: undefined >> undefined
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
Concept: undefined >> undefined
Boron fluoride exists as BF3 but boron hydride doesn’t exist as BH3. Give reason. In which form does it exist? Explain its structure.
Concept: undefined >> undefined
A nonmetallic element of group 13, used in making bullet proof vests is extremely hard solid of black colour. It can exist in many allotropic forms and has unusually high melting point. Its trifluoride acts as Lewis acid towards ammonia. The element exihibits maximum covalency of four. Identify the element and write the reaction of its trifluoride with ammonia. Explain why does the trifluoride act as a Lewis acid.
Concept: undefined >> undefined
Name the compounds whose line formulae are given below:

Concept: undefined >> undefined
Name the compounds whose line formulae are given below:
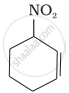
Concept: undefined >> undefined
Write structural formulae for compounds named as-
1-Bromoheptane
Concept: undefined >> undefined
Write structural formulae for compounds named as-
5-Bromoheptanoic acid
Concept: undefined >> undefined
Which of the following gases is not a green house gas?
Concept: undefined >> undefined
The gaseous envelope around the earth is known as atmosphere. The lowest layer of this is extended upto 10 km from sea level, this layer is ______.
Concept: undefined >> undefined
The acids present in acid rain are:
(i) Peroxyacetylnitrate
(ii) H2CO3
(iii) HNO3
(iv) H2SO4
Concept: undefined >> undefined
Green house effect leads to global warming. Which substances are responsible for green house effect?
Concept: undefined >> undefined
Acid rain is known to contain some acids. Name these acids and where from they come in rain?
Concept: undefined >> undefined
Some time ago formation of polar stratospheric clouds was reported over Antarctica. Why were these formed? What happens when such clouds break up by warmth of sunlight?
Concept: undefined >> undefined
Match the terms given in Column I with the compounds given in Column II.
| Column I | Column II |
| (i) Acid rain | (a) CHCl2 – CHF2 |
| (ii) Photochemical smog | (b) CO |
| (iii) Combination with haemoglobin | (c) CO2 |
| (iv) Depletion of ozone layer | (d) SO2 |
| (e) Unsaturated hydrocarbons |
Concept: undefined >> undefined
Match the pollutant(s) in Column I with the effect(s) in Column II.
| Column I | Column II |
| (i) Oxides of sulphur | (a) Global warming |
| (ii) Nitrogen dioxide | (b) Damage to kidney |
| (iii) Carbon dioxide | (c) ‘Blue baby’ syndrome |
| (iv) Nitrate in drinking water | (d) Respiratory diseases |
| (v) Lead | (e) Red haze in traffic and congested areas |
Concept: undefined >> undefined
