Please select a subject first
Advertisements
Advertisements
Read the passage given below:
| (1) | Ratan, a global brand in Dairy products, works on a business model popularly known as, 'The Ratan Model'. This model aims to provide value for money to the customers and protect the interests of farmers simultaneously. | ||||||||||
| (2) | The Ratan model is a three-tiered structure that is implemented in its Dairy production: Firstly, Ratan acts as a direct link between milk producers and consumers that removes the middlemen. Secondly, farmers (milk producers) control procurement, processing and marketing. Thirdly, it is a professionally managed organization. | ||||||||||
| (3) | One can understand the Ratan Model better by taking cognizance of 'Ratan's Target Audience', where it has targeted the mass market of India with no premium offerings and works on providing the best quality products at affordable prices. | ||||||||||
| (4) | So Ratan formulates its pricing policy on the low cost price strategy which has attracted a lot of customers in the past and it continues to do so. | ||||||||||
| (5) | Another stance used by Ratan's Target Audience is based on customer-wise targeting and industry wise targeting. This strategy divides the target audience on the following two bases : | ||||||||||
| (6) |
The above table showcases how Ratan has a diversified customer base. |
||||||||||
| (7) | Industry Based Target Audience: Ratan has segmented milk for various industries such as ice-cream manufacturers, restaurants, coffee shops, and many similar industries. Further, it has segmented butter, ghee and cheese for bakeries, snack retailers, confectioneries, and many more. | ||||||||||
| (8) | The target audience study tells us that Ratan has a strong presence in both Business to Business and Businessto-Customers. | ||||||||||
| (9) | Ratan's marketing campaigns and strategies are implemented in a very attractive way. For example, the story of the 'Ratan Girl' is a popular 'ad' icon. It is a hand drawn cartoon of a young girl. | ||||||||||
Based on your understanding of the passage answer any Six out of the Seven questions given below:
- What does 'The Ratan Model' aim at?
- In dairy production how many tiers are there?
- Ratan acts as a direct link...? Explain.
- "Ratan Target audience is described as a diversified market. Explain with reference to the given table.
- Name the two basis on which Ratan divides the target audience.
- In which two spheres does Ratan have a strong presence?
- Which is the most loved ad icon of Ratan?
Concept: Unseen Passage Comprehension
Read the passage given below:
| (1) | When we think of the game of cricket, we come to the conclusion that it is primarily a game that depends on outstanding physical activities, good hand-eye coordination, speed, skill and strength. It provides entertainment and generates strong feelings of excitement. A good match of cricket or of any other game neither adds to the existing stock of human knowledge nor reveals any secret of existence. It does not carry any deep meaning but most people, particularly the lover of sports attach deep emotions and numerous meanings to it. Games are thought of as a metaphor for life. They are supposed to teach many lessons. In fact, more is said and written about a cricket match than about scientific findings or great philosophy. |
| (2) | This is because games, like a morality play, in which settings and rules are made by us, can easily make people test their fair and foul conduct, principles of reward and punishment, and emotions of joy and disappointment. They can make us experience the thrill of war without exposing us to its dangers. A man watching a cricket match on T.V. and munching popcorn is like a surrogate warrior. In fact, games provide us with a safe outlet for our aggressiveness. If games become aggressive, they lose the very purpose of providing entertainment and purging us of our aggressiveness. They can calm our impatience without creating any conflict. |
| (3) | Commentators, journalists, politicians and analysts can do a great favour to the competing teams by keeping the excitement within limits. The teams should play without being dominated by feelings of national honour and shame. Excellent performance of the players of both teams should be enjoyed and appreciated. Winning or losing in a game should not be taken seriously. A game is fun if it is played with true spirit of sportsmanship. |
Based on your understanding of the passage, answer the questions given below:
- Complete the sentence by choosing an appropriate option: (1)
Most people conclude that cricket is primarily a game because ______.- it is played as a match
- it requires two teams
- it includes physical activity
- it depends only on skill and strength.
- Comment on the writer's reference to 'that cricket does not reveal any secret of existence. (1)
- List two responses to which watching a game of cricket gives rise to. (1)
- Select the option that conveys the opposite of 'destroy' from words used in the passage. (1)
- reveals
- experience
- generate
- purging
- The writer would not agree with the given statements based on paragraph 2, EXCEPT (1)
- Rules of any game are made by people.
- Watching a cricket match makes the viewer believe that he is fighting a battle.
- It is necessary for a game to be aggressive in order to build excitement.
- A game can test people's sense of fair judgement.
- With reference to the passage, a spectator is compared to a 'surrogate warrior'. (1)
Choose the option that best describes this phrase:- a spectator who is paid to watch.
- a spectator who is in pain while watching the match.
- a spectator who enjoys the match as an armchair soldier.
- a spectator who makes judgments about reward and punishment.
- Why does the writer compare games to a morality play? (1)
- Complete the given sentence with an appropriate inference with respect to the following: (1)
The writer says that games can calm our impatience without creating any conflict by ______. - The writer advises the players that games should not become aggressive because ______. (1)
- Select the most suitable title for the above passage. (1)
- Excellent Performance by Cricketers
- The Benefits of Playing Cricket
- Cricket - The King of Games
- The True Spirit of Playing Games
Concept: Unseen Passage Comprehension
Read the following report and answer the questions by choosing the correct/most appropriate option:
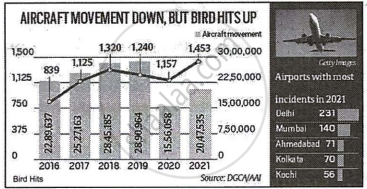 |
|
| (1) |
How bird hits happen, why they are a concern? New Delhi June 20 At least two bird strike incidents happened on Sunday. Both aircrafts returned to their airports of origin and were grounded for maintenance. An ABC aircraft, which took off from Guwahati towards Delhi, suffered damage to its left engine following a bird hit at 1600 feet. The pilots returned to Guwahati. The other strike happened on another flight from Patna to Delhi. The pilots of the aircraft suspected a bird strike during the take-off roll but continued to climb. Following the take-off rotation, they were informed by the cabin crew of sparks emanating from the left engine. Subsequently, the pilots were also informed by air traffic control of smoke coming out of one of the engines. The pilots declared an emergency and returned to Patna. |
| (2) |
Why are bird strikes a concern? Bird strikes are among the most common threats to aircraft safety, and they typically occur during take-off or landing. Dozens of bird-strikes happen each day but some can be more dangerous than others. Typically, when birds collide with an aircraft's airframe, it is unlikely to cause significant problems for the pilots flying. But there are instances when the aircraft engine ingests the birds. This can lead to a loss of thrust for the engine and cause manoeuvrability problems for the crew. In these cases, where a jet engine ingests a bird, procedures would generally call for pilots to land the plane at the closest airport. While most airframe bird strikes are not considered critical to air safety, if a collision cracks a window or a wind screen, pilots will look to land as early as possible. |
| (3) |
How critical are bird strikes to air safety? Smaller planes would generally be more susceptible to the dangers of bird strikes than larger ones. However, given that bird strikes mostly happen during take-off and landing, these incidents could distract the pilots during what are highly critical phases of flights that demand the complete attention of the crew. |
| (4) |
What causes bird-strikes? The presence of birds around an aircraft increases the chances of a bird strike. In the monsoon, as water puddles emerge on open grounds attracting insects to breed, the presence of birds increases. In some cases, bird hits also happen at higher altitudes when a plane is cruising. These are more dangerous than the low-altitude hits, given that they can cause rapid depressurization of cabins. Other reasons for bird activity around the airfield could be presence of landfills or waste disposal sites that can attract a large number of birds. |
- Does the following statement agree with the information given in paragraph 1? (1)
When an airplane is hit by a bird or is suspected to have been hit, the passengers must be asked to get down at once.
Select from the following:
True: If the statement agrees with the information.
False: If the statement contradicts the information.
Not Given: If there is no information on this. - Select the option that displays the most likely reason for bird hits. (1)
- when the aircraft is overloaded
- at the time of landing or taking off
- when the crew become negligent
- when the passengers become panicky
- Complete the sentence appropriately with one word. (1)
Smaller planes are generally more ______ than larger ones. - Complete the sentence by selecting the most appropriate option: (1)
When the window or the windscreen of the airplane is cracked due to a bird hit ______.- the crew must rush to the cockpit
- the pilot must land at the earliest airport
- the pilot must inform the maintenance engineer
- the pilot must get instructions from the aviation wing on what should do in emergency
- Based on the reading of the text, state a point to further the statement. (1)
Dozens of bird hits take place every day ______.
- but only a few are dangerous
- all of them are fatal
- but after every bird hit it is imperative to land the aircraft
- the pilot should ignore them
- Complete the sentence based on the following statement: (1)
Complete attention of the crew is demanded during take-off and landing.
We can say this because ______. - Complete the sentence appropriately with one/two words. (1)
In cases where the aircraft engine infests the bird, it leads to the loss of thrust and causes problems in ______. - Which are the areas more prone to bird hits? (1)
- Based on the reading of the text, state a point to challenge the given statement: (1)
The area around air fields should be clear of any waste disposal sites. - Look at the graph. It shows that between 2016 and 2021, whereas air flights have come down, the bird-hits have gone up. This implies that the incidence of bird hits has: (1)
- decreased
- increased
- remained constant
- been alarming
Concept: Unseen Passage Comprehension
Read the passage given below:
| (1) | Seagulls, as you know, never falter, never stall. To stall in the air is for them a disgrace and a dishonour. But Jonathan Livingston Seagull, unashamed, stretching his wings again in that trembling hard curve – slowing, slowing, and stalling once more –was no ordinary bird. Most gulls don't bother to learn more than the simplest facts of flight – how to get from shore to food and back again. For most gulls, it is not flying that matters, but eating. For this gull, though, it was not eating that mattered, but flight. More than anything else, Jonathan Livingston Seagull loved to fly. |
| (2) | This kind of thinking, he found, is not the way to make oneself popular with other birds. Even his parents were dismayed as Jonathan spent the whole day alone, making hundreds of low-level gliders, experimenting. "Why, Jon, why?" his mother asked. "Why is it so hard to be like the rest of the flock, Jon? Why can't you leave low flying to the pelicans, the albatross? Why don't you eat? Son, you're bone and feathers!" "I don't mind being bone and feathers, Mom. I just want to know what I can do in the air and what I can't, that's all. I just want to know." "See here Jonathan," said his father, not unkindly. "Winter isn't far away. Boats will be few, and the surface fish will be swimming deep. If you must study, then study food, and how to get it. This flying business is all very well, but you can't eat a glide, you know. Don't you forget that the reason you fly is to eat?" |
| (3) | Jonathan nodded obediently. For the next few days, he tried to behave like the other gulls; he really tried, screeching and fighting with the flock around the piers and fishing boats, diving on scraps of fish and bread. But he couldn't make it work. It wasn't long before Jonathan Gull was off by himself again, far out at sea, hungry, happy, learning. The subject was speed and in a week's practice he learned more about speed than the fastest gull alive. Time after time it happened. Careful as he was, working at the very peak of his ability, he lost control at a high speed. The key, he thought at last, dripping wet, must be to hold the wings still at high speeds – to flap up to fifty and then hold the wings still. |
| (4) | From two thousand feet he tried again, rolling into his dive, beak straight down, wings full out and stable from the moment he passed fifty miles per hour. It took tremendous strength, but it worked. In ten seconds he had blurred ninety miles per hour. Jonathan had set a world speed record for seagulls! But victory was short-lived. The instant he began his pullout, the instant he changed the angle of his wings, he snapped into the same uncontrollable disaster, and at ninety miles per hour, it hit him like dynamite. Jonathan Seagull exploded in midair and smashed down into a brick-hard sea. As he sank low in the water, a strange hollow voice sounded within him. There's no way around it. I am a seagull. I am limited by my nature. If I were meant to learn so much about flying, I'd have charts for brains. If I were meant to fly at speed, I'd have a falcon's short wings. Short wings. A falcon's short wings! That's the answer! What a fool I've been! All I need is a tiny little wing, all I need is to fold most of my wings and just fly on the tips along. Short wings! |
Based on your understanding of the passage, answer the questions given below:
- Complete the sentence by choosing an appropriate option: (1)
Majority of seagulls fly only short distances as ______.- they are more interested in food than flight
- they don't have energy
- they are not meant to fly low
- food is not available at high speed
- Why were Jonathan Livingston's parents' dismayed? (1)
- Give two reasons for Jonathan's unconventional behaviour. (1)
(Clue: think about Jonathan's point of view.) - Select the option that conveys the opposite of 'glory' from the words used in paragraph 1. (1)
- disgrace
- dishonour
- learning
- unashamed
- The writer would not agree with the given statements based on paragraph 2, EXCEPT (1)
- Jonathan could not fly but only glide.
- Jonathan wanted to be popular with other birds.
- Jonathan realised that even the albatross flew at high altitudes.
- The reason seagulls flew was to find food.
- Jonathan was different, from other seagulls. Based on your understanding of paragraph 2, list what Jonathan wanted to know. (1)
- What was the mother's concern about Jonathan? (1)
- Complete the given sentence with an appropriate inference with respect to the following: (1)
Father reminds Jonathan that he 'can't eat a glide' in order to ______. - It, wasn't long before Jonathan Gull was off by himself again, far out at sea. Which trait of Jonathan does this statement reveal? (1)
- practical bird
- persistent learner
- lonely and sad
- carefree and irresponsible
- Was it fair to fly like a falcon when he was just a seagull? Why does he say so? (1)
Concept: Unseen Passage Comprehension
Read the passage given below:
| (1) | Fast-Moving Consumer Goods (FMCG) are products that sell quickly at relatively low cost. FMCG is the fourth-largest sector in the Indian economy. There are three main segments in the sector – food and beverages, which accounts for 19% of the sector; healthcare, which accounts for 31% of the share; and household and personal care, which accounts for the remaining 50% share. The urban segment contributes to about 55% of the revenue share, while the rural segment accounts for 45%. Rise in rural consumption will drive the FMCG market. The Indian processed food market is projected to expand to US\[\$\] 470 billion by 2025, up from US\[\$\] 263 billion in 2019-20. |
| (2) | The Indian FMCG industry grew by 16% in 2021, a 9-year high, despite nationwide lockdowns, supported by consumption-led growth and value expansion from higher product prices, particularly for staples. Real household spending is projected to increase 9.1% after 2021, after a decrease of 9.3% in 2020 due to the economic impact of the pandemic. Price increases across product categories will offset the impact of rising raw material prices, along with volume growth and a resurgence of demand for discretionary items. |
| (3) | The FMCG sector has received good investments and support from the Government in the recent past. The sector witnessed healthy FDI inflows from April 2000-March 2022. Furthermore, as per the Union Budget 2022-23, a substantial amount has been allocated to the Department of Consumer Affairs, and an increased amount has been allocated to the Department of Food and Public Distribution. In 2021-22, the Government approved the Production-Linked Incentive Scheme for the Food Processing Industry (PLISFPI) with an outlay of a larger amount to help Indian brands of food products in the international markets. |
| (4) | The Government's Production-Linked Incentive (PLI) Scheme gives companies a major opportunity to boost exports. The future outlook of the FMCG rural sector looks on track now. Rural consumption has increased, led by a combination of increasing income and higher aspiration levels. There is an increased demand for branded products in rural India. The growth of the organised sector in FMCG is expected to rise with an increased level of brand consciousness, augmented by the growth in modem retail. |
 |
|
| Table. FMCG Products | |
| 1. Processed foods: | Cheese products, cereals |
| 2. Prepared meals: | Ready-to-eat meals |
| 3. Beverages: | Bottled water, aerated drinks, and juices |
| 4. Baked goods: | Biscuits, bread |
| 5. Fresh foods, frozen foods, and dry goods: | Fruits, vegetables, milk, butter, frozen food, and nuts |
| 6. Medicines: | Aspirin, pain relievers, and over-the-counter medication that can be purchased without a prescription |
| 7. Cleaning products: | Baking soda, washing powder |
| 8. Cosmetics and toiletries: | Beauty products, soaps, toothpaste |
| 9. Office supplies: | Pens, pencils |
Based on your understanding of the passage, answer the questions given below:
- Does the following statement agree with the information given in para 1? (1)
Food and beverages segment dominates the Global FMCG market and is expected to retain its dominance.
Select from the following:- True - If the statement agrees with the information.
- False - If the statement contradicts the information.
- Not Given - If there is no information on this.
- What do you think gives opportunities to boost exports? (1)
- Select the option that displays the most likely reason for FMCG rural sector being on track. (1)
- FMCG sector has been unable to get investments from the government.
- PLI schemes don't reach the rural sector.
- Gross Merchandise Value (GMV) of the online grocery segment in India is expected to double in the next five years.
- There is a desire to buy branded products in the villages too.
- Complete the sentence based on the following statement: (1)
The Indian FMCG industry grew by 16% in 2021, a 9-year high, despite nationwide lockdowns because ______. - From the given pie chart, which segment of FMCG accounts for the maximum percentage of the sector? (1)
- Complete the given sentence by selecting the most appropriate option: (1)
The combination of increasing income and higher aspiration levels in the rural areas has led to ______.- the Government taking a direct interest
- the demand for branded products
- price increases across product categories
- the fall of supply in urban areas
- How is the Government trying to help Indian brands of food products in the international markets? (1)
- Complete the given sentence by selecting the most appropriate option: (1)
The concluding paragraph of the passage makes a clear case ______.- that the rural people also want branded products
- that the FMCG industry will not focus on brand consciousness
- for the role of data analytics in the FMCG industry
- for the Government's role by providing incentives
- What will be the impact of the increased level of brand consciousness? (1)
- Based on the reading of the passage, correct the following statement: (1)
The urban segment contributes to about 45% of the revenue share, while the rural segment accounts for 55%.
Concept: Unseen Passage Comprehension
Read the following approaches of human geography and choose the correct option of the chronological order:
- Exploration and description
- Spatial organisation
- Areal differentiation
- Regional analysis
Concept: Nature of Human Geography
Study the given statement I and II and choose the correct option.
Statement-I: A society which fails to acknowledge and take effective measures to remove such discriminations cannot be treated as a civilised one.
Statement-II: Duly acknowledging the adverse impacts of these discriminations, launched a-campaign called 'Beti Bachao-Beti Padhao' in India.
Concept: Promoting Gender Sensitivity
Which among the followings is NOT the correct feature of rural settlements?
Concept: Types of Settlements >> Concept of Rural Settlements
Choose the correct pair of the following types of towns:
Concept: Urbanisation in India
Which one of the following is garrison cantonment town?
Concept: Urbanisation in India
"Broadly 90 percent of the world population lives in about 10 percent of its land area." Examine the statement in context of patterns of population distribution in the world.
Concept: Patterns of Population Distribution in the World
On the given political outline Map of India, locate and label any five of the following geographical features with appropriate symbols:
- State with the highest level of urbanisation.
- State with the lowest level of density of population
- The leading coffee producing state
- Khetri - copper mines
- Jamnagar - Oil refinery
- The Northern terminal station of North-South Corridor.
- Tuticorin - major sea-port
Concept: Urbanisation in India
Who, among the following economists created the Human Development Index in the year 1990?
Concept: Growth and Development
Which one of the following approaches was initially proposed by the International Labour Organisation (ILO)?
Concept: Approaches of Human Development
Which of the following is NOT a key area in human development?
Concept: Growth and Development
Read the following statements regarding open-cast mining and choose the correct option:
- This is the cheapest way of mining.
- The output is in large quantity.
- It requires specially designed lifts, drills.
- It requires special ventilation system.
Concept: Primary Occupations >> Mining
Which among the following is NOT the mineral based industry?
Concept: Household Industries Or Cottage Manufacturing
Explain any five factors affecting the location of industries in the world.
Concept: Manufacturing
Under which one of the following sectors, collection of information comes?
Concept: Quaternary Activities
In a developed economy, the majority of workers get employment in which of the following sectors of economy?
Concept: Quaternary Activities
