Advertisements
Advertisements
प्रश्न
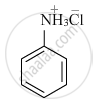
Write the structures of main products when aniline reacts with the following reagents : HCl
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Why does NH3 act as a Lewis base?
Arrange the following:
In increasing order of basic strength:
Aniline, p-nitroaniline and p-toluidine
Write the structures of the main products of the following reactions:

The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
Account for the following:
Acylation of aniline is carried out in the presence of pyridine.
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
A Solution of methyl amine shows which type of property with litmus paper?
Which of the following compound cannot be produced if 1-propane amine is treated with NaNO2 and HCl?
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
