English Medium
Academic Year: 2025-2026
Date & Time: 25th February 2026, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contains 39 questions. All questions are compulsory.
- The question paper is divided into three sections - A, B and C.
Section A: Biology (30 Marks)
Section B: Chemistry (25 Marks)
Section C: Physics (25 Marks) - The question paper has MCQs, VSAs, SAs, LAs and CBQs. Marks are given against each question.
- There are case based questions (CBQs) with three sub-questions and are of 4 marks each.
- Divide your answer sheet into three sections as per the question paper -
Section A (Biology), Section B (Chemistry) and Section C (Physics).
It is compulsory to answer each question in its respective section. Do not mix answers of one section into the other section. - Instructions are given with each section and question, wherever necessary.
- Kindly note that a separate question paper has been provided for visually impaired candidates.
- There is no overall choice in the question paper. However, an internal choice has been provided in a few questions. Only one of the choices in such questions must be attempted.
The carbohydrates, which are not used immediately in plants, are stored in the form of ‘a’, which serves as an internal energy reserve. What does ‘a’ stand for?
Starch
Sugar
Fat
Protein
Chapter:
Spirogyra, despite being a multi-cellular organism, still shows a simple reproductive method. Select the correct option with regard to reproduction in Spirogyra:
Asexual reproduction by division of cells into two cells.
Sexual reproduction by division of cells into many cells.
Asexual reproduction by breaking up of Spirogyra filaments into smaller parts
Sexual reproduction by the formation of gametes after meiosis of cells of filaments followed by fusion of male and female gametes.
Chapter:
Identify the type of tropic movement that is responsible for the movement of the pollen tube towards an ovule from the following options.
Hydrotropism
Geotropism
Chemotropism
Phototropism
Chapter:
The genotype of green stemmed tomato plant is denoted by ‘GG’ and that of purple stemmed tomato plant as ‘gg’. Hybrid of a cross between these two were self-pollinated to obtain F2 progeny. What will be the ratio of GG, Gg and gg in this F2 progeny?
2 : 1 : 1
3 : 1 : 0
1 : 1 : 2
1 : 2 : 1
Chapter:
Select the option that represents the correct balanced equation for the process of photosynthesis:
\[\ce{CO2 + O2 + H2O ->[Chlorophyll] C6H12O6 + H2O}\]
\[\ce{6CO2 + 6H2O ->[Chlorophyll][Sunlight] C6H12O6 + O2}\]
\[\ce{CO2 + H2O ->[Sunlight] C6H12O6}\]
\[\ce{6CO2 + 12H2O ->[Chlorophyll][Sunlight] C6H12O6 + 6O2 + 6H2O}\]
Chapter:
Which of the following limits the number of trophic levels in a food chain?
Availability of water
Deficient food supply
Decrease in energy at higher levels
Pollution in air
Chapter:
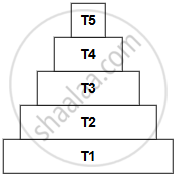
In the given figure, identify the trophic level having maximum concentration of harmful chemicals.
T1
T5
T4
T3
Chapter:
Assertion (A): Blood plasma transports carbon dioxide in dissolved form, while oxygen is transported by respiratory pigments.
Reason (R): Carbon dioxide is more soluble in water than in oxygen.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): There are generally a greater number of individuals at the lower trophic levels of an ecosystem.
Reason (R): The greatest number is of the producers.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
“The timing and the amount of hormone secreted by a gland are regulated in the human body.” Justify this statement with the help of an example.
Chapter:
Electrical impulse is an excellent means to transmit information in the animal body, but there are some limitations to the use of electrical impulses. State any two limitations.
Chapter:
Do all variations in a species have equal chances of surviving in the environment in which they find themselves? Explain.
Chapter:
Suggest one precaution to be taken before disposing of the following:
Sewage from homes
Chapter:
Suggest one precaution to be taken before disposing of the following:
Industrial wastes
Chapter:
Draw a neat diagram of the human respiratory system and label its lungs, trachea, bronchi and alveoli.
Chapter:
State the role of the following in the process of respiration:
Alveoli
Chapter:
State the role of the following in the process of respiration:
Respiratory pigments
Chapter:
Giving two examples, justify the statement that the sex of the offspring in all animals is not always genetically determined.
Chapter:
Differentiate between the sex chromosomes of male and female human beings.
Chapter:
Read the following passage and answer the questions that follow:
| A middle-aged person is facing some cognitive changes in himself. His early symptoms included mild forgetfulness. For example, forgetting where he left his mobile or failing to find words to complete his sentences. He was facing problems in driving a car, climbing up and down the stairs, inserting a plug into an outlet. He slowly developed a sad mood and anxiety. |
(a) What are voluntary actions? [1]
(b) Which part of the brain is responsible for precision of voluntary actions? [1]
(c) (i) Explain the role of the medulla. [2]
OR
(c) (ii) How do the animal muscles move? [2]
Chapter:
Name an organ that helps in the formation of male germ cells in humans and also mention the condition that favours the formation of male germ cells.
Chapter:
In male reproductive system, write the pathway followed to deliver male germ cells to the site of fertilisation.
Chapter:
Advertisements
Mention the main physical feature of the male germ cell that helps it move towards the female germ cell.
Chapter:
Radha has three children. She does not want to have more children. Radha visits a nearby hospital, to know the methods to prevent pregnancy.
- Mention the name of any two ways other than the surgical methods, following which Radha can avoid unwanted pregnancies.
- Name the surgical methods to prevent pregnancy.
- Give one example each of sexually transmitted diseases caused by bacteria and virus.
Chapter:
Tomato is a natural source of which acid?
Acetic acid
Tartaric acid
Oxalic acid
Citric acid
Chapter:
Which of the following compounds is used for removing permanent hardness of water?
Bleaching powder
Washing soda
Baking soda
Caustic soda
Chapter:
Which of the following are decomposition reactions?
- \[\ce{2KClO3 -> 2KCl + 3O2}\]
- \[\ce{MgO + H2O -> Mg(OH)2}\]
- \[\ce{2H2O -> H2 + O2}\]
- \[\ce{Zn + CuSO4 -> ZnSO4 + Cu}\]
(i) and (ii)
(iii) and (iv)
(i) and (iii)
(ii) and (iv)
Chapter:
A metal carbonate reacts with a solution X which forms a salt, water and a gas Y. What are X and Y?
X - Sodium hydroxide, Y - Carbon dioxide
X - Sodium hydroxide, Y - Hydrogen
X - Hydrochloric acid, Y - Carbon dioxide
X - Hydrochloric acid, Y - Hydrogen
Chapter:
When ferrous sulphate crystals are heated in a test tube, we observe that ______.
A colourless gas with no smell is evolved.
A brown gas is evolved.
The green colour of the salt fades and a gas with the smell of burning sulphur is evolved.
The green colour of the salt fades and no gas is evolved.
Chapter:
A student notices that her silver jewellery turned dull and had a grey-black coating over it after wearing for a few months. What results in the change in colour of the silver metal?
The polish over the jewellery was removed after wearing for a few months.
The jewellery comes in contact with air, moisture and acids and corrodes.
Dust particles over the jewellery change its colour.
Its colour changes due to rusting.
Chapter:
When Mg metal is treated with hot water, it starts floating on the surface of the water. The reason for its different behaviour is:
Mg is a lighter element than water.
Mg reacts with water to produce H2 gas which helps in floating.
Mg reacts with water to produce N2 gas which helps in floating.
Mg reacts with water to form CO2 gas which helps in floating.
Chapter:
Assertion (A): Carbon shows the property of catenation.
Reason (R): This is due to strong C–C bond.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Write equations for the reactions of iron with steam.
Chapter: [3] Metals and Non-metals
What happens when calcium reacts with water? Write the chemical equation of the reaction of calcium with water.
Chapter: [3] Metals and Non-metals
Name a metal which is so soft that it can be cut with a knife.
Chapter:
Name a non-metal which is a good conductor of electricity.
Chapter:
Using the electronic configuration, explain how Mg atom combines with oxygen to form magnesium oxide by transfer of electrons.
[Atomic number: Mg = 12, O = 8]
Chapter:
Give a reason for the following:
Carbonate and sulphide ores are usually converted into oxides during the process of extraction of metals.
Chapter:
Give reasons:
Aluminium oxide is considered as an amphoteric oxide.
Chapter:
Give reasons:
Metals like Na, K, Ca and Mg are never found in their free state in nature.
Chapter:
Define corrosion.
Chapter: [1] Chemical Reactions and Equations
Define the following term:
Double displacement reaction
Chapter:
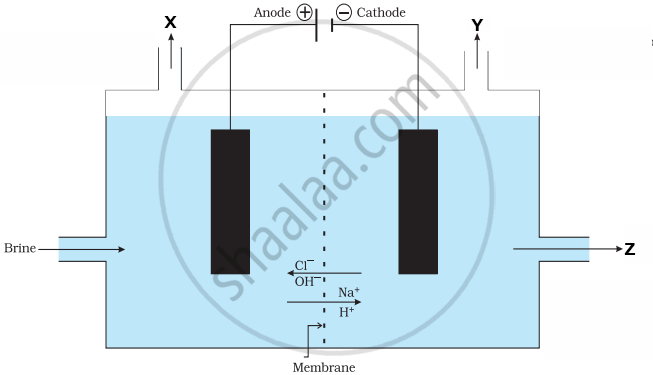
Read the following passage and answer the questions that follow:
|
In the given diagram, when electricity is passed through an aqueous solution of a common salt, a substance ‘Z’ is produced, along with the evolution of gases ‘X’ and ‘Y’. When a burning matchstick is brought near the gas ‘Y’, it burns with a pop sound, whereas ‘X’ is used for disinfecting drinking water. When ‘X’ is passed through slaked lime, an insoluble substance ‘A’ is produced.
|
(a) Write the names of the gases ‘X’ and ‘Y’. [1]
(b) Write the balanced chemical equation for the formation of substance ‘Z’. [1]
(c) (i) What happens: [2]
- If a drop of red litmus solution is added to the aqueous solution of substance ‘Z’?
- If CO2 gas is passed through ammoniacal solution of aqueous NaCl?
OR
(c) (ii) Identify the substance ‘A’ and write the balanced chemical equation of the reaction which takes place in its preparation. [2]
Chapter:
Advertisements
A carbon compound ‘P’ of molecular formula C2H6O is found to be neutral when tested with red and blue litmus. A gas that burns with a pop sound is produced when sodium metal reacts with compound ‘P’.
- Identify the compound ‘P’. [1]
- The carbon compound ‘P’ is heated with conc. H2SO4 at 443 K to produce compound ‘Q’. [2]
- Write the chemical equation for the reaction.
- What is the role of conc. H2SO4 in this reaction?
- What is likely to be observed on heating compound ‘P’ with ethanoic acid with an acid as a catalyst? Write the chemical equation for the reaction. [2]
Chapter:
State the reason why carbon can neither form C4+ cation nor C4− anion.
Chapter:
What type of bond is formed between carbon and hydrogen?
Chapter:
How is ethanoic acid prepared from ethanol? Give the chemical equation.
Chapter: [4] Carbon and its Compounds
Complete the following chemical equation:
\[\ce{CH3COOC2H5 + NaOH ->}\]
Chapter: [4] Carbon and its Compounds
From the following options choose the one which gives negative magnification greater than one:
Concave mirror; Nature of image - real
Concave mirror; Nature of image - virtual
Convex mirror; Nature of image - real
Convex mirror; Nature of image - virtual
Chapter:
A thin beam of white light falls on a face of a glass prism and a spectrum is obtained on a screen. Which of the following options is the correct sequence of colours from top to bottom on the screen?
Red, Green, Yellow, Violet, Blue
Red, Yellow, Green, Blue, Violet
Violet, Blue, Green, Yellow, Red
Violet, Red, Green, Yellow, Blue
Chapter:
Assertion (A): Direction of force exerted on a current-carrying conductor placed in a magnetic field remains same if the directions of both magnetic field and current flowing through the conductor are reversed.
Reason (R): The direction of force exerted on a current-carrying conductor placed in an external magnetic field can be determined by using magnetic compass.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
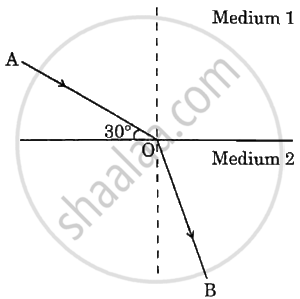
With the help of above given ray diagram, calculate. the angle of refraction. Given that the refractive index of Medium 2 with respect to Medium 1 is `sqrt 3`.
Chapter:
How does the focal length of eye lens change as the distance of the object from the eyes is altered? Explain.
Chapter:
Why is the concave lens used as a corrective lens for a myopic eye?
Chapter:
Write the name of the lens and position of the object in front of it so that a virtual and magnified image is formed.
Chapter:
An object is placed perpendicular to the principal axis of a convex lens of focal length 20 cm. If object is located at 30 cm from this lens, using lens formula find out the position and nature of the image formed.
Chapter:
How is scattering of light different from reflection of light? Explain.
Chapter:
The values of current I flowing in a given resistor for the corresponding values of potential difference V applied across the ends of resistor are given below in the table:
| I (Amperes) | 0.4 | 0.8 | 1.2 | 1.6 | 2.0 |
| V (Volts) | 1.2 | 2.4 | 3.6 | 4.8 | 6.0 |
Plot a graph between V and I and calculate the resistance of that resistor.
Chapter:
Read the following passage and answer the questions that follow:
| Swati, a class 10 student, observes that when she passes close to the refrigerator in her kitchen, she feels the heat, although the things kept inside the refrigerator are cool. |
(a) Describe the cause of heating in the above-mentioned case. [1]
(b) A current I flows through a resistor of resistance R when the potential difference across it is V. Applying Ohm’s law, write the formula for amount of heat produced by the resistor in time t. [1]
(c) (i) Write any two practical applications of heating effect of electric current. [2]
OR
(c) (ii) Define the commercial unit of electric energy and express it in Joules (J). [2]
Chapter:
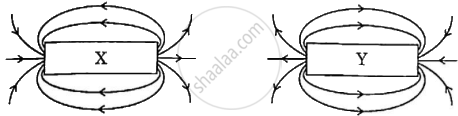
The magnetic field lines due to two bar magnets X and Y are shown in the given diagram. If the magnet X is displaced towards magnet Y, will there be repulsion or attraction between them? Justify.
Chapter:
A metallic conductor is suspended perpendicular to the magnetic field of a horseshoe magnet. The conductor gets displaced towards the left when a current is passed through it. What will be the effect on displacement of conductor if the:
- Current through the conductor is increased?
- Horseshoe magnet is replaced by another stronger horseshoe magnet?
- Direction of current through the conductor is reversed?
Chapter:
Draw a schematic diagram of a domestic electric circuit which includes the following devices:
- A fuse
- A power meter
- One light point
- A power plug
Chapter:
Why is it necessary to connect an earth wire to the electrical appliances having metallic covers?
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2025 - 2026
Previous year Question paper for CBSE Class 10 Science-2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.