Advertisements
Advertisements
प्रश्न
Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Advertisements
उत्तर
Pentaamminecarbonatocobalt(III) chloride
APPEARS IN
संबंधित प्रश्न
Write IUPAC names of the following compounds:

Complete the following reactions
NH3+3Cl2(excess) ---->
Write the structures of compounds A, B and C in the following reactions
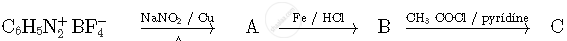
Write structures of compounds A and B of the following reaction :

The coordination number of the central ion may be obtained from:
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Which of the following is an ionic ligand?
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
