Advertisements
Advertisements
प्रश्न
What happens to the atomic number of element when (i) An α -particle, (ii) A β -particle 1 and (iii) γ-radiation is emitted?
Advertisements
उत्तर
(i) α particle is represented by 2He4 this means it contains 2 protons so after emitting a α particle atomic number of element would decrease bv 2 unit.
(ii) β particle is represented by -1θ° after emitting a β particle atomic number of element would increase bv 1 unit.
(iii) Y radiations have no charge and no mass so after emitting a Y radiation atomic number of element remain unchanged.
APPEARS IN
संबंधित प्रश्न
When is said to be the nucleus unstable?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
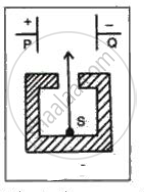
Why is the source S placed in a thick lead container?
A radioactive nuclide `""_86^226` Ra decays by emission of two alpha particles, one beta particle and gamma rays. Which of the following is the resulting nuclide X?
What are the uses of radioactive isotopes in following?
(a) Medical field,
(b) Agriculture,
(c) Industries?
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
Name two radioactive substances.
An electrons emitter must have how much work function and melting point.
A radioctive sample decays `7/8` times its original quantity in 15 minutes. The half-life of the sample is ______.
Assertion (A): The force of repulsion between atomic nucleus and α-particle varies with distance according to inverse square law.
Reason (R): Rutherford did α-particles scattering experiment.
