Advertisements
Advertisements
प्रश्न
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
Why is the source S placed in a thick lead container?
Advertisements
उत्तर
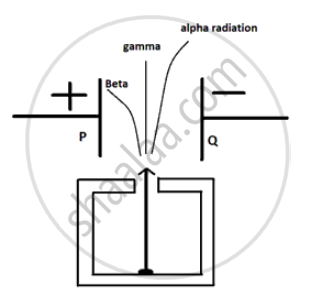
(i) Alpha particles are heavy in mass and are positively charged so they are deflected less and are deflected towards - charged plate.
(ii) Beta particles are negIigible in mass so they are highly deflected by electric field anc they are negatively charge particle so they are deflected towards positive charged plate.
(iii) Gamm a radiations have no mass and no charge so they are not deflected by electric field.
Source S is placed in thick lead container because it is radioactive substance and radiates. Thick walls of lead absorb a II the radiation except radiation going straight outside towards opened end. Thus, thick walIs help to reduce leakage of radiations outside.
APPEARS IN
संबंधित प्रश्न
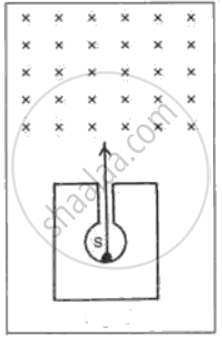
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
A nucleus of an element X which has the symbol `""_84^202` X emits an alpha particle and then a beta particle. The final nucleus is `""_"b"^"a"` Y Find a and b.
What happens to the position of an element in the periodic table when it emits an alpha particle
In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
Name the three main parts of a Cathode Ray Tube.
Mention four properties of Becquerel rays.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
When an α-particle of mass m moving with velocity ν bombards on a heavy nucleus of charge Ze, its distance of closest approach from the nucleus depends on m as:
