Advertisements
Advertisements
प्रश्न
Obtain the relation between the decay constant and half life of a radioactive sample.
Advertisements
उत्तर
The number of atoms at any instant in a radioactive sample is given by
N=N0e−λt
where
N=total number of atoms at any instant
N0=number of atoms in radioactive substance at t=0
λ=decay constant
t=time
When t=T (Where T is the half life of the sample)
`N=N_0/2`
`=>N_0/2=N_0e^(-lambdat)`
`=1/2=e^(-lambdaT)`
`=>e^(lambdaT)=2`
Taking log on both the sides, we get
`lambdaT=log_e2=2.303 lod_10 2`
`=>T=(2.303 lod_10 2)/lambda`
`=>T=(2.303 xx 0.3010)/lambda`
`=>T=0.6931/lambda`
Thus, half life of a radioactive substance is inversely propotional to decay constant.
APPEARS IN
संबंधित प्रश्न
State the law of radioactive decay.
The Q value of a nuclear reaction A + b → C + d is defined by
Q = [mA+ mb − mC − md]c2 where the masses refer to the respective nuclei. Determine from the given data the Q-value of the following reactions and state whether the reactions are exothermic or endothermic.
\[\ce{^12_6C + ^12_6C ->^20_10Ne + ^4_2He}\]
Atomic masses are given to be
`"m"(""_1^2"H")` = 2.014102 u
`"m"(""_1^3"H")` = 3.016049 u
`"m"(""_6^12C)` = 12.000000 u
`"m"(""_10^20"Ne")` = 19.992439 u
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
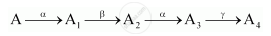
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
A freshly prepared radioactive source of half-life 2 h emits radiation of intensity which is 64 times the permissible safe level. The minimum time after which it would be possible to work safely with this source is
28Th emits an alpha particle to reduce to 224Ra. Calculate the kinetic energy of the alpha particle emitted in the following decay:
`""^228"Th" → ""^224"Ra"^(∗) + alpha`
`""^224"Ra"^(∗) → ""^224"Ra" + γ (217 "keV")`.
Atomic mass of 228Th is 228.028726 u, that of 224Ra is 224.020196 u and that of `""_2^4H` is 4.00260 u.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Obtain a relation between the half-life of a radioactive substance and decay constant (λ).
Define one Becquerel.
What percentage of radioactive substance is left after five half-lives?
Draw a graph showing the variation of decay rate with number of active nuclei.
The activity R of an unknown radioactive nuclide is measured at hourly intervals. The results found are tabulated as follows:
| t (h) | 0 | 1 | 2 | 3 | 4 |
| R (MBq) | 100 | 35.36 | 12.51 | 4.42 | 1.56 |
- Plot the graph of R versus t and calculate the half-life from the graph.
- Plot the graph of ln `(R/R_0)` versus t and obtain the value of half-life from the graph.
