Advertisements
Advertisements
प्रश्न
Moving from left to right, the size of the atom decreases.
पर्याय
Right
Wrong
Advertisements
उत्तर
Moving from left to right, the size of the atom decreases- Right
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Which is greater in size Fe2+ or Fe3+?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Give reason for the following:
Argon atom is bigger than chlorine atom.
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
Nanometer unit is used to measure atomic radius.
The size of an atom depends on the number of valence electrons.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Which of the following is the correct order of size?
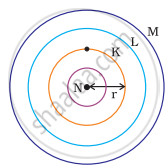
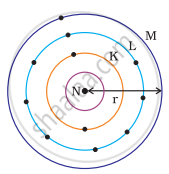
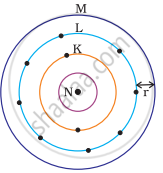
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
Which one of the following has the largest atomic radius?