Advertisements
Advertisements
प्रश्न
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Advertisements
उत्तर
Size of the atoms Decrease from left to right across a period and increase on descending in a group of normal elements.
APPEARS IN
संबंधित प्रश्न
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Give reason for the following:
Argon atom is bigger than chlorine atom.
Arrange the following in order of increasing radii:
CI- , CI
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
_______ is the distance between the nucleus of the atom and its outermost shell.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
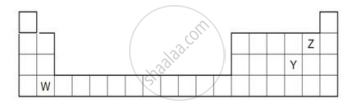
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)
