Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Advertisements
उत्तर
Na > Mg > Si > S > Cl
APPEARS IN
संबंधित प्रश्न
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Arrange the following in order of increasing radii:
CI- , CI
Write a scientific reason.
Atomic radius goes on increasing down a group.
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
Atomic radius is expressed in the unit _______.
Write an Explanation.
Atomic radius
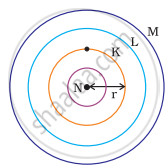
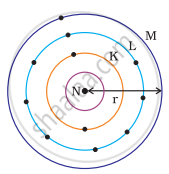
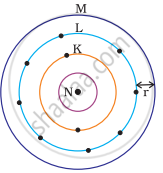
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I