Advertisements
Advertisements
प्रश्न
In a given sample, two radioisotopes, A and B, are initially present in the ration of 1 : 4. The half lives of A and B are respectively 100 years and 50 years. Find the time after which the amounts of A and B become equal.
Advertisements
उत्तर
Let NA be the concentration of A after tA time and NB be the concentration of B after tB time.
So, NA = N0e−λAtA
NB = 4N0e−λBtB (as N0B = 4N0A)
Now half-life of A is 100 years and B is 50 years.
`So lambda_A = (ln^2)/100 and lambda_B = (ln^2)/50`
Dividing we get
`(lambda_A)/(lambda_B)= 1/2 or lambda_B =2lambda_A`
Now let after t years NA = NB
So`(N_A)/(N_B) = e^(-lambdaA')/(4e^(lambdaB')`
`N_A =N_B`
`4e^(-lambdaB') =e^(-lambdaA') `
`4= e^-(lambda_A -lambda_B)`
`ln4 = -(+lambda_A - 2lambda_A)t (because lambda_B = 2lambda_A)`
`ln4 = lambda_At`
`t = (ln4)/(ln2) xx 100 (because lambda_A = (ln2)/100)`
= 200 years
संबंधित प्रश्न
The decay constant of radioactive substance is 4.33 x 10-4 per year. Calculate its half life period.
State the law of radioactive decay.
A radioactive nucleus 'A' undergoes a series of decays as given below:

The mass number and atomic number of A2 are 176 and 71 respectively. Determine the mass and atomic numbers of A4 and A.
The radioactive isotope D decays according to the sequence
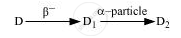
If the mass number and atomic number of D2 are 176 and 71 respectively, what is (i) the mass number (ii) atomic number of D?
In a radioactive decay, neither the atomic number nor the mass number changes. Which of the following particles is emitted in the decay?
Before the year 1900 the activity per unit mass of atmospheric carbon due to the presence of 14C averaged about 0.255 Bq per gram of carbon.
(a) What fraction of carbon atoms were 14C?
(b) An archaeological specimen containing 500 mg of carbon, shows 174 decays in one hour. What is the age of the specimen, assuming that its activity per unit mass of carbon when the specimen died was equal to the average value of the air? The half-life of 14C is 5730 years.
After 1 hour, `(1/8)^"th"` of the initial mass of a certain radioactive isotope remains undecayed. The half-life of the isotopes is ______.
Suppose we consider a large number of containers each containing initially 10000 atoms of a radioactive material with a half life of 1 year. After 1 year ______.
Sometimes a radioactive nucleus decays into a nucleus which itself is radioactive. An example is :
\[\ce{^38Sulphur ->[half-life][= 2.48h] ^{38}Cl ->[half-life][= 0.62h] ^38Air (stable)}\]
Assume that we start with 1000 38S nuclei at time t = 0. The number of 38Cl is of count zero at t = 0 and will again be zero at t = ∞ . At what value of t, would the number of counts be a maximum?
The radioactivity of an old sample of whisky due to tritium (half-life 12.5 years) was found to be only about 4% of that measured in a recently purchased bottle marked 10 years old. The age of a sample is ______ years.
