Advertisements
Advertisements
प्रश्न
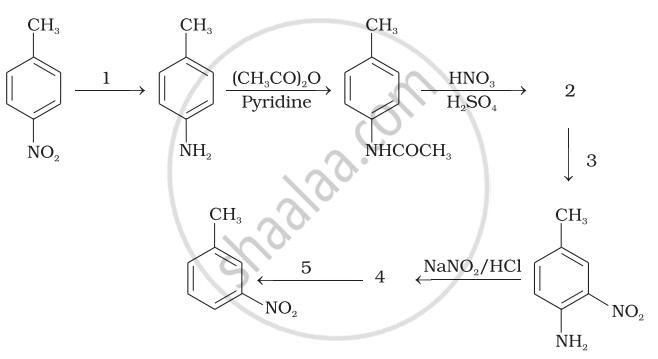
Illustrate the following reaction giving suitable example in each case: Diazotisation
Write the reactions involved in Diazotisation
Advertisements
उत्तर १

उत्तर २
The process of the formation of the diazonium salt by the reaction of the aromatic amine with the nitrous acid is known as the diazotization reaction

APPEARS IN
संबंधित प्रश्न
Write equations of the following reactions:
Acetylation of aniline
Arenium ion involved in the bromination of aniline is:
(i)

(ii)

(iii)

(iv)

Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
How will you carry out the following conversions?
toluene `->` p-toluidine
Predict the reagent or the product in the following reaction sequence.
Which of the following statement about primary amines is false?
In order to distinguish between C2H5NHz and C6H5NHz, which of the following reagents is useful?
Aniline when treated with cone. HNO3 gives
Benzene diazonium chloride is a ______.
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
