Advertisements
Advertisements
प्रश्न
Depict the galvanic cell in which the reaction \[\ce{Zn(s) + 2Ag+(aq) → Zn^{2+}(aq) + 2Ag(s)}\] takes place. Further show:
- Which of the electrode is negatively charged?
- The carriers of the current in the cell.
- Individual reaction at each electrode.
रासायनिक समीकरणे/रचना
लघु उत्तर
Advertisements
उत्तर
The set-up will be similar to as shown below:
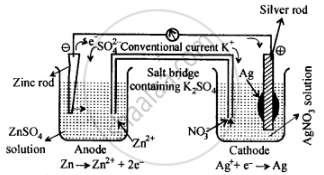
The cell will be represented as:
\[\ce{Zn(s) | Zn^{2+}(aq) || Ag^+(aq) | Ag(s)}\]
- Anode, i.e., zinc electrode, will be negatively charged.
- The current will flow from silver to zinc in the external circuit.
- At anode: \[\ce{Zn(s) -> Zn^{2+}(aq) + 2e–}\]
At cathode: \[\ce{Ag+(aq) + e– -> Ag(s)}\]
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
