Advertisements
Advertisements
प्रश्न
Complete flow chart given below.
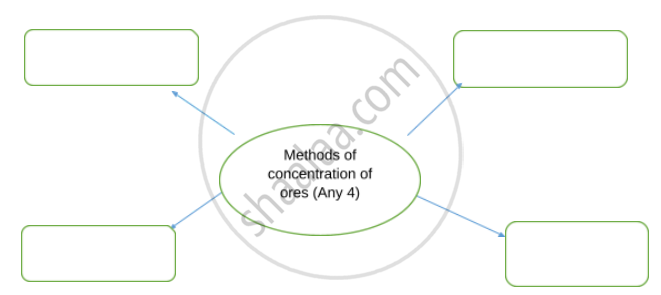
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: carbonates
Name the processes involved in refining of ores.
Explain the following term:
Gangue
Explain the following terms:
smelting
`ZnCO_3` ..............................
..............................
Why is flux used in the blast furnace?
Which metal is used for:
lithographic plates for printing
What is magnalium? Name the main elements present in it? Write its one use ?
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
The chemical formula of zinc blend is _______.
Write the molecular formulae of the following compound.
Ferrous tungstate
Distinguish between:
Froth floatation - Leaching
Explain concept with example/explain with the help of a balanced equation.
Minerals
Observe the figure and name and explain in brief the following method:

Observe the figure and name and explain in brief the following method:

In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
After partial roasting, the sulphide of copper is reduced by ______.
Name the process that is employed to refine aluminium.
Identify false statement regarding lithium aluminium hydride?
