Advertisements
Advertisements
प्रश्न
Explain concept with example/explain with the help of a balanced equation.
Minerals
Advertisements
उत्तर
- Most metals being reactive do not occur in nature in the free state but are found in a combined state as their salts such as oxides, carbonates, sulphides, and nitrates.
- However, some unreactive metals like silver, gold, platinum, generally occur in a free state.
- The compounds of metals that occur in nature along with the impurities are called minerals.
- For example, bauxite is a mineral of aluminium.
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Identify A and B and give their reactions with dil HCL and dil H2SO4 ?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: nitrates
Explain the following terms:
(a) flux
`2Pb(NO3)_2`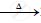 .........................
.........................
state the reactions at the two electrodes ?
Which metal is used for:
lithographic plates for printing
Which metal is used for:
making face creams
State two properties of brass that render it more useful for some purpose than its components.
Arrange the metals in (2) in the decreasing order of reactivity.
Name the following:
Name an allotrope of a non-metal that allows electricity to pass through it.
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Explain the hydraulic separation method with a neat labelled diagram.
The chemical name of rust is ______.
Which of the following methods is used to separate wolframite and stannic oxide present in cassiterite?
Bassemerisation is used in the extraction of ______.
Which of following metals occurs in native state?
A process of extracting metals from aqueous solutions of their salts using suitable reducing agents is called ______
Explain the following term:
Metallurgy
Calcination is used in metallurgy for the removal of?
