Advertisements
Advertisements
प्रश्न
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Advertisements
उत्तर
| Group A | Group B |
| 1. ZnS | d) Zinc blend |
| 2. HgS | c) Cinnabar |
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Hydrogen
Explain the following terms:
smelting
Name the methods used for purification?
`Cu (OH)2` ................................
................................
`ZnCO_3`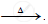 ..............................
..............................
`2Cu_2O +Cu_2S` ......................
......................
Which metal is used for:
making pipes, buckets, water tanks,
What is magnalium? Name the main elements present in it? Write its one use ?
Complete the following flow chart and answer the questions below:

i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
In Wilfley table method to separate particles of gangue _______ method is used.
The chemical formula of zinc blend is _______.
Distinguish between:
Froth floatation - Leaching
Explain concept with example/explain with the help of a balanced equation.
Gangue
Observe the figure and name and explain in brief the following method:

The chemical name of rust is ______.
Which of following metals occurs in native state?
Calcination is used in metallurgy for the removal of?
