Advertisements
Advertisements
प्रश्न
Match the columns.
| Group A | Group B |
| 1. ZnS | a) Copper Sulphide |
| 2. HgS | b) Bauxite |
| c) Cinnabar | |
| d) Zinc blend |
Advertisements
उत्तर
| Group A | Group B |
| 1. ZnS | d) Zinc blend |
| 2. HgS | c) Cinnabar |
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Give the importance of the following for living beings:
Hydrogen
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
Name:
(b) two metallic oxides which cannot be reduced by carbon, carbon monoxide or hydrogen
Explain the following terms:
(c) slag
Why is flux used in the blast furnace?
What is magnalium? Name the main elements present in it? Write its one use ?
Complete the following flow chart and answer the questions below:

i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
What do you observe when hydrogen is passed over heated copper oxide?
Write the molecular formulae of the following compound.
Copper pyrite
Explain the hydraulic separation method with a neat labelled diagram.
Identify the following method of concentration of ores and explain briefly.
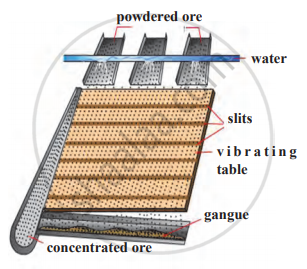
Complete the following flowchart.

Which of the following methods is used to separate wolframite and stannic oxide present in cassiterite?
Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
Explain the following term:
Metallurgy
