Advertisements
Advertisements
प्रश्न
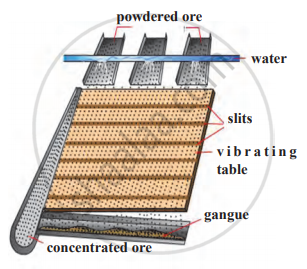
Explain the froth floatation method with a neat labelled diagram.
Advertisements
उत्तर
- The froth floatation process is based on two opposite properties of the particles, hydrophilic and hydrophobic.
- The metal sulphide particles are hydrophobic. Due to this property, they get wetted mainly with oil. On the other hand, gangue particles are hydrophilic and get wetted by water.
- In this method, the finely divided metal ore is added in a big tank containing large amount of water. To this, certain vegetable oil (pine oil, eucalyptus oil, etc.) is added for the formation of froth.
- In the tank, compressed air is circulated through the water. Bubbles are formed due to this blown air. A rotating agitator at the centre of the floatation tank agitates the mixture and draws air into it to form bubbles. As a result, foam is formed which carries metal sulphide particles. The foam rises to the surface of water and floats. Therefore, this method is called froth floatation process.
- This method is used to separate metal sulphide ores such as zinc blende (ZnS) and copper pyrite (CuFeS2).

Froth floatation method
APPEARS IN
संबंधित प्रश्न
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
Explain the following term:
Gangue
Explain the following terms:
smelting
`Ag2O` ................................
................................
`ZnCO_3` ..............................
..............................
`HgS + O_2` ..................
..................
Which metal is used for:
lithographic plates for printing
What do you observe when hydrogen is passed over heated copper oxide?
Cassiterite is a copper ore.
Draw a neat and labelled diagram for Froth Floatation Process.
Explain concept with example/explain with the help of a balanced equation.
Minerals
Identify the following method of concentration of ores and explain briefly.
Chemical formula of rust is ______
State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
Zirconium is refined by ____________.
Which of the following methods is used to separate wolframite and stannic oxide present in cassiterite?
A process of extracting metals from aqueous solutions of their salts using suitable reducing agents is called ______
Explain the following term:
Metallurgy
