Advertisements
Advertisements
प्रश्न
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + Na2SO3 -> MnO2 + Na2SO4 + KOH}\]
Advertisements
उत्तर
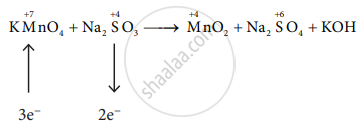
\[\ce{2KMnO4 + 3Na2SO3 -> MnO2 + Na2SO4 + KOH}\]
\[\ce{2KMnO4 + 3Na2SO3 -> 2MnO2 + 3Na2SO4 + KOH}\]
\[\ce{2KMnO4 + 3Na2SO3 + H2O -> 2MnO2 + 3Na2SO4 + 2KOH}\]
APPEARS IN
संबंधित प्रश्न
1 g of an impure sample of magnesium carbonate (containing no thermally decomposable impurities) on complete thermal decomposition gave 0.44 g of carbon dioxide gas. The percentage of impurity in the sample is
What do you understand by the term oxidation number?
Distinguish between oxidation and reduction.
Balance the following equation by the oxidation number method.
\[\ce{K2Cr2O7 + Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + I2 + H2O}\]
Balance the following equation by the oxidation number method.
\[\ce{Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + H2C2O4 + H2SO4 -> K2SO4 + MnSO4 + CO2 + H2O}\]
Reducing agent is a species that ____________.
In the redox reaction, \[\ce{2MnO^-_4 + SO^{2-}_3 + H2O -> 2MnO^{2-}_4 + SO^{2-}_4 + 2H^+}\], the reductant is:
In the cell represented by \[\ce{Pb(s) | Pb^{2+} (1 M) || Ag+ (1 M) | Ag(s)}\], the reducing agent is ______.
Which is the best oxidizing agent among the following?
