Advertisements
Advertisements
प्रश्न
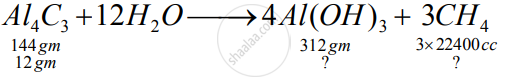
Aluminium carbide reacts with water according to the following equation :
`Al_4C_3 + 12H_2O-> 4Al(OH)_3 + 3CH_4`
1)What mass of aluminium hydroxide is formed from 12g of aluminium carbide?
2) What volume of methane at s.t.p. is obtained from 12g of aluminium carbide?
[Relatively molecular weight of `Al_4Cl_3 = 144; Al(OH)_3 = 78]`
Advertisements
उत्तर
1)
So, the amount of 3 Al(OH)3 formed will be 26 gm
2) From 12 gm Al4C3 5600 cc methane will be formed
APPEARS IN
संबंधित प्रश्न
Prove the Following :
2 X V.D. = Molecular mass.
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of oxygen formed when 5.05g of potassium nitrate decomposes completely.
(K = 39, 0 = 16, N = 14)
Calculate the percentage of water in ferrous sulphate crystals.
[Fe = 56, S = 32, O =16, H = 1].
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
A compound 'X' consists of 4.8% of C and 95.2% of Br by mass.
Name the type of chemical reaction by which X can be prepared from ethane.
4.5 moles of calcium carbonate are reacted with dilute hydrochloric acid.
- Write the equation for the reaction.
- What is the mass of 4.5 moles of calcium carbonate? (Relative molecular mass of calcium carbonate is 100).
- What is the volume of carbon dioxide liberated at STP?
- What mass of calcium chloride is formed? (Relative molecular mass of calcium chloride is 111).
- How many moles of HCl are used in this reaction?
Explain the term:
Vapour density
Calculate the relative molecular mass of:
CHCl3
Find the weight of 0.5 mole of O2.
Calculate the mass of nitrogen supplied to soil by 5 kg of urea [CO(NH2)2].
[O = 16; N = 14; C = 12; H = 1]
