Advertisements
Advertisements
प्रश्न
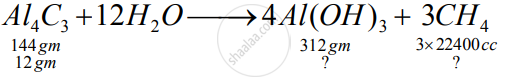
Aluminium carbide reacts with water according to the following equation :
`Al_4C_3 + 12H_2O-> 4Al(OH)_3 + 3CH_4`
1)What mass of aluminium hydroxide is formed from 12g of aluminium carbide?
2) What volume of methane at s.t.p. is obtained from 12g of aluminium carbide?
[Relatively molecular weight of `Al_4Cl_3 = 144; Al(OH)_3 = 78]`
Advertisements
उत्तर
1)
So, the amount of 3 Al(OH)3 formed will be 26 gm
2) From 12 gm Al4C3 5600 cc methane will be formed
APPEARS IN
संबंधित प्रश्न
10g of NaCl solution is mixed with 17g of silver nitrate solution. Calculate the weight of silver chloride precipitated.
AgNO3 + NaCl → AgCI + NaNO3
Calculate the percentage of phosphorous in the fertilizer superphosphate, Ca(H2PO4)2. [Ca = 40, H =1, P =31, O = 16] (Correct to 1 decimal place)
A metal M, forms a volatile chloride containing 65.5% Chlorine. If the density of the chloride relative to hydrogen is 162.5, find the molecular formula of the chloride. [M = 56, Cl = 35.5]
When heated, potassium permanganate decomposes according to the following equation:
\[\ce{2KMnO4 -> \underset{solid residue}{K2MnO4 + MnO2} + O2}\]
Given that the molecular mass of potassium permanganate is 158 g, what volume of oxygen (measured at room temperature) would be obtained by the complete decomposition of 15.8 g of potassium permanganate? (Molar volume at room temperature is 24 litres). [K = 39, Mn = 55, O = 16]
Calculate the percentage of sodium in sodium aluminium fluoride (Na3AIF6).
[F = 19, Na = 23, Al = 27]
A gaseous hydrocarbon contains 82.76% of carbon. Given that its vapour density is 29, find its molecular formula.
[C = 12, H = 1]
Give two tests of the following:
Oxygen
Calculate the volume occupied at S.T.P. by 2 moles of SO2.
The following question refers to one mole of chlorine gas.
If the relative atomic mass of chlorine is 35.5, what will be the mass of 1 mole of chlorine gas?
67.2 litres of hydrogen combines with 44.8 litres of nitrogen to form ammonia under specific conditions as:
\[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\]
Calculate the volume of ammonia produced. What is the other substance, if any, that remains in the resultant mixture?
