Advertisements
Advertisements
A hydrogen atom emits ultraviolet radiation of wavelength 102.5 nm. What are the quantum numbers of the states involved in the transition?
Concept: undefined >> undefined
(a) Find the first excitation potential of He+ ion. (b) Find the ionization potential of Li++ion.
Concept: undefined >> undefined
Advertisements
A group of hydrogen atoms are prepared in n = 4 states. List the wavelength that are emitted as the atoms make transitions and return to n = 2 states.
Concept: undefined >> undefined
Find the maximum Coulomb force that can act on the electron due to the nucleus in a hydrogen atom.
Concept: undefined >> undefined
A hydrogen atom in a state having a binding energy of 0.85 eV makes transition to a state with excitation energy 10.2 e.V (a) Identify the quantum numbers n of the upper and the lower energy states involved in the transition. (b) Find the wavelength of the emitted radiation.
Concept: undefined >> undefined
Whenever a photon is emitted by hydrogen in Balmer series, it is followed by another photon in Lyman series. What wavelength does this latter photon correspond to?
Concept: undefined >> undefined
A hydrogen atom in state n = 6 makes two successive transitions and reaches the ground state. In the first transition a photon of 1.13 eV is emitted. (a) Find the energy of the photon emitted in the second transition (b) What is the value of n in the intermediate state?
Concept: undefined >> undefined
What is the energy of a hydrogen atom in the first excited state if the potential energy is taken to be zero in the ground state?
Concept: undefined >> undefined
In a photo diode, the conductive increases when the material is exposed to light. It is found that the conductivity changes only if the wavelength is less than 620 nm. What is the band gap?
(Use Planck constant h = 4.14 × 10-15 eV-s, Boltzmann constant k = 8·62 × 10-5 eV/K.)
Concept: undefined >> undefined
A gas of hydrogen-like ions is prepared in a particular excited state A. It emits photons having wavelength equal to the wavelength of the first line of the Lyman series together with photons of five other wavelengths. Identify the gas and find the principal quantum number of the state A.
Concept: undefined >> undefined
Find the maximum angular speed of the electron of a hydrogen atom in a stationary orbit.
Concept: undefined >> undefined
Suppose, in certain conditions only those transitions are allowed to hydrogen atoms in which the principal quantum number n changes by 2. (a) Find the smallest wavelength emitted by hydrogen. (b) List the wavelength emitted by hydrogen in the visible range (380 nm to 780 nm).
Concept: undefined >> undefined
Find the mutual inductance between the straight wire and the square loop of figure.
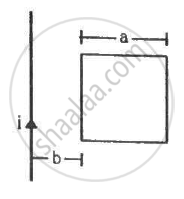
Concept: undefined >> undefined
Find the mutual inductance between the circular coil and the loop shown in figure.
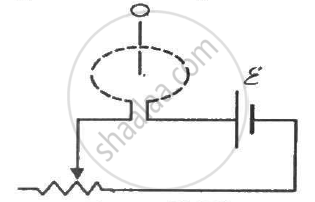
Concept: undefined >> undefined
The current in a long solenoid of radius R and having n turns per unit length is given by i= i0 sin ωt. A coil having N turns is wound around it near the centre. Find (a) the induced emf in the coil and (b) the mutual inductance between the solenoid ant the coil.
Concept: undefined >> undefined
A solenoid of length 20 cm, area of cross-section 4.0 cm2 and having 4000 turns is placed inside another solenoid of 2000 turns having a cross-sectional area 8.0 cm2 and length 10 cm. Find the mutual inductance between the solenoids.
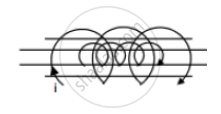
Concept: undefined >> undefined
The average kinetic energy of molecules in a gas at temperature T is 1.5 kT. Find the temperature at which the average kinetic energy of the molecules of hydrogen equals the binding energy of its atoms. Will hydrogen remain in molecular from at this temperature? Take k = 8.62 × 10−5 eV K−1.
Concept: undefined >> undefined
Find the temperature at which the average thermal kinetic energy is equal to the energy needed to take a hydrogen atom from its ground state to n = 3 state. Hydrogen can now emit red light of wavelength 653.1 nm. Because of Maxwellian distribution of speeds, a hydrogen sample emits red light at temperatures much lower than that obtained from this problem. Assume that hydrogen molecules dissociate into atoms.
Concept: undefined >> undefined
Average lifetime of a hydrogen atom excited to n = 2 state is 10−8 s. Find the number of revolutions made by the electron on the average before it jumps to the ground state.
Concept: undefined >> undefined
Show that the ratio of the magnetic dipole moment to the angular momentum (l = mvr) is a universal constant for hydrogen-like atoms and ions. Find its value.
Concept: undefined >> undefined
