Advertisements
Advertisements
प्रश्न
Average lifetime of a hydrogen atom excited to n = 2 state is 10−8 s. Find the number of revolutions made by the electron on the average before it jumps to the ground state.
Advertisements
उत्तर
Frequency of electron (f) is given by
`f = (me^4)/(4∈_0^2n^3h^3)`
Time period is given by
` T = 1/f`
`T = (4∈_0^2n^3h^3)/me^`
Here,
h = Planck's constant
m = Mass of the electron
e = Charge on the electron
`∈_0`= Permittivity of free space
`∴ T =(4xx(8.85xx10^-12)xx(2)^3xx(6.63xx10^-34)^3)/((9.10xx10^-31)xx(1.6xx106^-16)^4 `
`= 12247.735xx10^-19 S`
Average life time of hydrogen, `t = 10^-3 S`
Number of revolutions is given by
`N = t/T`
`rArr N=(10^-8)/(12247.735xx10^-19)`
N = 8.2 × 105 revolution
APPEARS IN
संबंधित प्रश्न
Find the wavelength of the electron orbiting in the first excited state in hydrogen atom.
Which wavelengths will be emitted by a sample of atomic hydrogen gas (in ground state) if electrons of energy 12.2 eV collide with the atoms of the gas?
The minimum orbital angular momentum of the electron in a hydrogen atom is
Which of the following curves may represent the speed of the electron in a hydrogen atom as a function of trincipal quantum number n?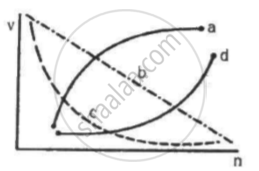
A hydrogen atom in ground state absorbs 10.2 eV of energy. The orbital angular momentum of the electron is increased by
Which of the following products in a hydrogen atom are independent of the principal quantum number n? The symbols have their usual meanings.
(a) vn
(b) Er
(c) En
(d) vr
Let An be the area enclosed by the nth orbit in a hydrogen atom. The graph of ln (An/A1) against ln(n)
(a) will pass through the origin
(b) will be a straight line with slope 4
(c) will be a monotonically increasing nonlinear curve
(d) will be a circle
Calculate the smallest wavelength of radiation that may be emitted by (a) hydrogen, (b) He+ and (c) Li++.
A hydrogen atom emits ultraviolet radiation of wavelength 102.5 nm. What are the quantum numbers of the states involved in the transition?
A group of hydrogen atoms are prepared in n = 4 states. List the wavelength that are emitted as the atoms make transitions and return to n = 2 states.
A gas of hydrogen-like ions is prepared in a particular excited state A. It emits photons having wavelength equal to the wavelength of the first line of the Lyman series together with photons of five other wavelengths. Identify the gas and find the principal quantum number of the state A.
Suppose, in certain conditions only those transitions are allowed to hydrogen atoms in which the principal quantum number n changes by 2. (a) Find the smallest wavelength emitted by hydrogen. (b) List the wavelength emitted by hydrogen in the visible range (380 nm to 780 nm).
Electrons are emitted from an electron gun at almost zero velocity and are accelerated by an electric field E through a distance of 1.0 m. The electrons are now scattered by an atomic hydrogen sample in ground state. What should be the minimum value of E so that red light of wavelength 656.3 nm may be emitted by the hydrogen?
A hydrogen atom moving at speed υ collides with another hydrogen atom kept at rest. Find the minimum value of υ for which one of the atoms may get ionized.
The mass of a hydrogen atom = 1.67 × 10−27 kg.
When a photon is emitted from an atom, the atom recoils. The kinetic energy of recoil and the energy of the photon come from the difference in energies between the states involved in the transition. Suppose, a hydrogen atom changes its state from n = 3 to n = 2. Calculate the fractional change in the wavelength of light emitted, due to the recoil.
The Balmer series for the H-atom can be observed ______.
- if we measure the frequencies of light emitted when an excited atom falls to the ground state.
- if we measure the frequencies of light emitted due to transitions between excited states and the first excited state.
- in any transition in a H-atom.
- as a sequence of frequencies with the higher frequencies getting closely packed.
Positronium is just like a H-atom with the proton replaced by the positively charged anti-particle of the electron (called the positron which is as massive as the electron). What would be the ground state energy of positronium?
In the Auger process an atom makes a transition to a lower state without emitting a photon. The excess energy is transferred to an outer electron which may be ejected by the atom. (This is called an Auger electron). Assuming the nucleus to be massive, calculate the kinetic energy of an n = 4 Auger electron emitted by Chromium by absorbing the energy from a n = 2 to n = 1 transition.
