Advertisements
Advertisements
An electric immersion heater is rated 1250 W. Calculate the time in which it will heat 20 kg of water at 5°C to 65°C.
Concept: undefined >> undefined
Derive an expression for finding out the specific heat capacity of a body (solid) from the readings of an experiment given below:
(i) Mass of empty calorimeter (with stirrer) = m1 gm
(ii) Mass of the metal piece = M gm
(iii) Mass of colorimeter and water = m2 gm
(iv) Initial temperature and water = t1°C
(v) Temperature of hot solid (metal piece) = t2 °C
(vi) Final temperature of the mixture = t°C
(vii) Specific heat of calorimeter = 0.4 J gm / °C
Concept: undefined >> undefined
Advertisements
The diagram below shows a cooling curve for 200 g of water. The heat is extracted at the rate of 100 Js-1. Answer the questions that follow:

- Calculate specific heat capacity of water.
- Heat released in the region BC.
Concept: undefined >> undefined
Two metals A and B have specific heat capacities in the ratio 2 : 3. If they are supplied the same amount of heat then
Which metal piece will show a greater rise in temperature given their masses is the same?
Concept: undefined >> undefined
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
Which metal piece will have greater mass if the rise in temperature is the same for both metals?
Concept: undefined >> undefined
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
If the mass ratio of metal A and metal B is 3:5 then calculate the ratio in which their temperatures rise.
Concept: undefined >> undefined
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
If specific heat capacity of metal A is 0.26 Jg-1 0C-1 then calculate the specific heat capacity of metal B.
Concept: undefined >> undefined
Why is water used as a coolant in radiators of a car?
Concept: undefined >> undefined
Specific heat capacity of a substance X is 1900 Jkg-1°C-1 means ______.
Concept: undefined >> undefined
Thermal capacities of substances A and B are same. If mass of A is more than mass of B then:
Which substance will have more specific heat capacity?
Concept: undefined >> undefined
Calculate the distance covered by the Ultrasonic wave having a velocity of 1.5 kms-1 in 14 s, when it is received after reflection by the receiver of the SONAR.
Concept: undefined >> undefined
Name the class of the lever shown in the picture below:
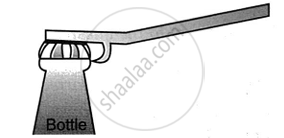
Concept: undefined >> undefined
A ______ lever will always have M.A. > 1.
Concept: undefined >> undefined
Give one reason for the use of ultrasonic waves in echo depth sounding.
Concept: undefined >> undefined
Classify the following into levers:
Claw hammer
Concept: undefined >> undefined
Classify the following into levers:
Sugar tongs
Concept: undefined >> undefined
State the principle on which SONAR is based.
Concept: undefined >> undefined
Two blocks P and Q of different metals having their mass in the ratio 2 : 1 are given same amount of heat. Their temperature rises by same amount. Compare their specific heat capacities.
Concept: undefined >> undefined
Give one example where high specific heat capacity of water is used as heat reservoir.
Concept: undefined >> undefined
