Please select a subject first
Advertisements
Advertisements
Assertion: \[\ce{SF6}\] cannot be hydrolysed but \[\ce{SF4}\] can be.
Reason: Six \[\ce{F}\] atoms in \[\ce{SF6}\] prevent the attack of \[\ce{H2O}\] on sulphur atom of \[\ce{SF6}\].
Concept: undefined >> undefined
\[\begin{array}{cc}
\ce{CH3 - C ≡ CH ->[40{%} H2SO4][1{%} HgSO4] A ->[Isomerisation] CH3 - C - CH3}\\
\phantom{........................................}||\\
\phantom{........................................}\ce{O}\\
\end{array}\]
Structure of ‘A’ and type of isomerism in the above reaction are respectively.
Concept: undefined >> undefined
Advertisements
Arenium ion involved in the bromination of aniline is:
(i)

(ii)

(iii)

(iv)

Concept: undefined >> undefined
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Concept: undefined >> undefined
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Concept: undefined >> undefined
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
Concept: undefined >> undefined
Why is aniline soluble in aqueous HCl?
Concept: undefined >> undefined
How will you carry out the following conversions?
toluene `->` p-toluidine
Concept: undefined >> undefined
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Concept: undefined >> undefined
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Concept: undefined >> undefined
A colourless substance ‘A’ \[\ce{(C6H7N)}\] is sparingly soluble in water and gives a water soluble compound ‘B’ on treating with mineral acid. On reacting with \[\ce{CHCl3}\] and alcoholic potash ‘A’ produces an obnoxious smell due to the formation of compound ‘C’. Reaction of ‘A’ with benzenesulphonyl chloride gives compound ‘D’ which is soluble in alkali. With \[\ce{NaNO2}\] and \[\ce{HCl}\], ‘A’ forms compound ‘E’ which reacts with phenol in alkaline medium to give an orange dye ‘F’. Identify compounds ‘A’ to ‘F’.
Concept: undefined >> undefined
Predict the reagent or the product in the following reaction sequence.
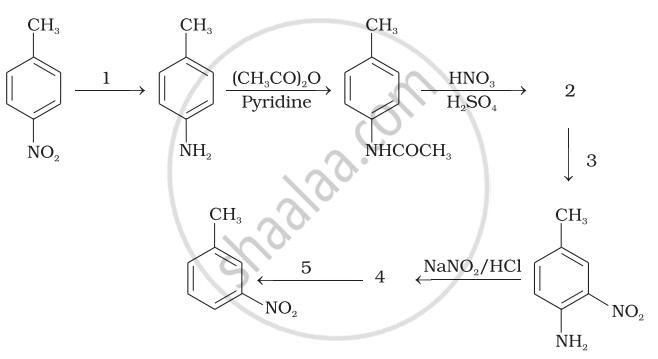
Concept: undefined >> undefined
Discuss briefly, giving an example of the role of coordination compounds in biological systems.
Concept: undefined >> undefined
Account for the following:
Ti(IV) is more stable than the Ti (II) or Ti(III).
Concept: undefined >> undefined
Draw the structures of the given compound - 3-Bromo-4-phenylpentanoic acid
Concept: undefined >> undefined
What is meant by the chelate effect? Give an example.
Concept: undefined >> undefined
Draw the structure of the following compound.
3-Bromo-4-phenylpentanoic acid
Concept: undefined >> undefined
What is meant by the chelate effect? Give an example.
Concept: undefined >> undefined
Draw the structure of the following compound.
3-Bromo-4-phenylpentanoic acid
Concept: undefined >> undefined
What is meant by the chelate effect? Give an example.
Concept: undefined >> undefined
