Advertisements
Advertisements
How will you carry out the following conversions?
toluene `->` p-toluidine
Concept: undefined >> undefined
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Concept: undefined >> undefined
Advertisements
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Concept: undefined >> undefined
A colourless substance ‘A’ \[\ce{(C6H7N)}\] is sparingly soluble in water and gives a water soluble compound ‘B’ on treating with mineral acid. On reacting with \[\ce{CHCl3}\] and alcoholic potash ‘A’ produces an obnoxious smell due to the formation of compound ‘C’. Reaction of ‘A’ with benzenesulphonyl chloride gives compound ‘D’ which is soluble in alkali. With \[\ce{NaNO2}\] and \[\ce{HCl}\], ‘A’ forms compound ‘E’ which reacts with phenol in alkaline medium to give an orange dye ‘F’. Identify compounds ‘A’ to ‘F’.
Concept: undefined >> undefined
Predict the reagent or the product in the following reaction sequence.
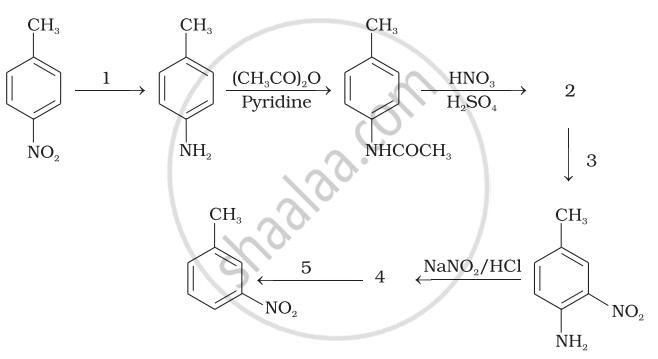
Concept: undefined >> undefined
Proteins are found to have two different types of secondary structures viz. α-helix and β-pleated sheet structure. α-helix structure of protein is stabilised by:
Concept: undefined >> undefined
Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?
Concept: undefined >> undefined
Optical rotations of some compounds along with their structures are given below which of them have D configuration.
| (I) |  |
| (II) |  |
| (III) |  |
Concept: undefined >> undefined
Proteins can be classified into two types on the basis of their molecular shape i.e., fibrous proteins and globular proteins. Examples of globular proteins are:
(i) Insulin
(ii) Keratin
(iii) Albumin
(iv) Myosin
Concept: undefined >> undefined
In fibrous proteins, polypeptide chains are held together by:
(i) van der Waals forces
(ii) disulphide linkage
(iii) electrostatic forces of attraction
(iv) hydrogen bonds
Concept: undefined >> undefined
Which of the following are purine bases?
(i) Guanine
(ii) Adenine
(iii) Thymine
(iv) Uracil
Concept: undefined >> undefined
α-Helix is a secondary structure of proteins formed by twisting of polypeptide chain into right-handed screw like structures. Which type of interactions are responsible for making the α-helix structure stable?
Concept: undefined >> undefined
Protein found in a biological system with a unique three-dimensional structure and biological activity is called a native protein. When a protein in its native form, is subjected to a physical change like change in temperature or a chemical change like, change in pH, denaturation of protein takes place. Explain the cause.
Concept: undefined >> undefined
Which moieties of nucleosides are involved in the formation of phosphodiester linkages present in dinucleotides? What does the word diester in the name of linkage indicate? Which acid is involved in the formation of this linkage?
Concept: undefined >> undefined
Assertion: β-glycosidic linkage is present in maltose,

Reason: Maltose is composed of two glucose units in which C–1 of one glucose unit is linked to C–4 of another glucose unit.
Concept: undefined >> undefined
Explain the terms primary and secondary structure of proteins. What is the difference between α-helix and β-pleated sheet structure of proteins?
Concept: undefined >> undefined
Which of the following is not a semisynthetic polymer?
Concept: undefined >> undefined
The commercial name of polyacrylonitrile is ______.
Concept: undefined >> undefined
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Concept: undefined >> undefined
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Concept: undefined >> undefined
