Advertisements
Advertisements
Would you prefer a material with a high work-function or a low work-function to be used as a cathode in a diode?
Concept: undefined >> undefined
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
Concept: undefined >> undefined
Advertisements
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Concept: undefined >> undefined
Why does thermionic emission not take place in non-conductors?
Concept: undefined >> undefined
The cathode of a diode valve is replaced by another cathode of double the surface area. Keeping the voltage and temperature conditions the same, will the place current decrease, increase or remain the same?
Concept: undefined >> undefined
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?

Concept: undefined >> undefined
The anode of a thermionic diode is connected to the negative terminal of a battery and the cathode to its positive terminal.
Concept: undefined >> undefined
The constant A in the Richardson−Dushman equation for tungsten is 60 × 104 A m−2K−2. The work function of tungsten is 4.5 eV. A tungsten cathode with a surface area 2.0 × 10−5 m2 is heated by a 24 W electric heater. In steady state, the heat radiated by the heater and the cathode equals the energy input by the heater and the temperature becomes constant. Assuming that the cathode radiates like a blackbody, calculate the saturation current due to thermions. Take Stefan's Constant = 6 × 10−8 W m−2 K−1. Assume that the thermions take only a small fraction of the heat supplied.
Concept: undefined >> undefined
Define the term "Threshold frequency", in the context of photoelectric emission.
Concept: undefined >> undefined
Define the term wavefront. Using Huygen’s wave theory, verify the law of reflection.
Concept: undefined >> undefined
Answer the following question.
Three photodiodes D1, D2, and D3 are made of semiconductors having band gaps of 2.5 eV, 2 eV, and 3 eV respectively. Which of them will not be able to detect light of wavelength 600 nm?
Concept: undefined >> undefined
Answer the following question.
Under what conditions is the phenomenon of total internal reflection of light observed? Obtain the relation between the critical angle of incidence and the refractive index of the medium.
Concept: undefined >> undefined
Answer the following question.
Three lenses of focal length +10 cm, —10 cm and +30 cm are arranged coaxially as in the figure given below. Find the position of the final image formed by the combination.

Concept: undefined >> undefined
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
Concept: undefined >> undefined
Answer the following question.
A charged particle q is moving in the presence of a magnetic field B which is inclined to an angle 30° with the direction of the motion of the particle. Draw the trajectory followed by the particle in the presence of the field and explain how the particle describes this path.
Concept: undefined >> undefined
Answer the following question.
Explain briefly how Rutherford scattering of α-particle by a target nucleus can provide information on the size of the nucleus.
Concept: undefined >> undefined
Answer the following question.
Define the term wavefront. Using Huygen's wave theory, verify the law of reflection.
Concept: undefined >> undefined
The work function of aluminum is 4⋅2 eV. If two photons each of energy 2⋅5 eV are incident on its surface, will the emission of electrons take place? Justify your answer.
Concept: undefined >> undefined
Define a wavefront. Using 'Huygens' principle, draw the shape of a refracted wavefront, when a plane wave is incident on a convex lens.
Concept: undefined >> undefined
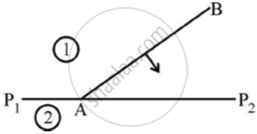
A plane wave front AB propagating from denser medium (1) into a rarer medium (2) is incident on the surface P1P2 separating the two media as shown in fig.
Using Huygen’s principle, draw the secondary wavelets and obtain the refracted wave front in the diagram.
Concept: undefined >> undefined
