Please select a subject first
Advertisements
Advertisements
Draw a formula for the first five members of the homologous series beginning with the given compound.
H–COOH
Concept: undefined >> undefined
Draw a formula for the first five members of the homologous series beginning with the given compound.
CH3COCH3
Concept: undefined >> undefined
Advertisements
Draw a formula for the first five members of the homologous series beginning with the given compound.
H–CH=CH2
Concept: undefined >> undefined
Identify the functional group in the following compound.
Concept: undefined >> undefined
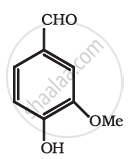
Identify the functional group in the following compound.
Concept: undefined >> undefined
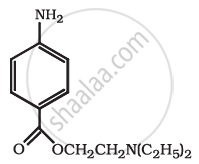
Identify the functional group in the following compound.

Concept: undefined >> undefined
Complete the following reaction:
\[\ce{H_{2(g)} + M_M O_{O(s)} ->[\Delta]}\] ?
Concept: undefined >> undefined
Complete the following reaction:
\[\ce{CO_{(g)} + H_{2(g)} ->[\Delta][catalyst]}\] ?
Concept: undefined >> undefined
Complete the following reaction:
\[\ce{C3H_{8(g)} + 3H2O_{(g)} ->[\Delta][catalyst]}\]?
Concept: undefined >> undefined
Complete the following reaction:
\[\ce{Zn_{(s)} + NaOH_{(aq)} ->[heat]}\]?
Concept: undefined >> undefined
An aqueous solution of borax is _______.
Concept: undefined >> undefined
Explain the Structure of Diborane.
Concept: undefined >> undefined
What do you mean by ozone hole?
Concept: undefined >> undefined
Write Lewis structure of the following compounds and show formal charge on atom.
\[\ce{HNO3}\]
Concept: undefined >> undefined
Write Lewis structure of the following compounds and show formal charge on atom.
\[\ce{NO2}\]
Concept: undefined >> undefined
Write Lewis structure of the following compounds and show formal charge on atom.
\[\ce{H2SO4}\]
Concept: undefined >> undefined
Represent diagrammatically the bond moments and the resultant dipole moment in \[\ce{CO2, NF3}\] and \[\ce{CHCl3}\].
Concept: undefined >> undefined
Which of the following reactions increases production of dihydrogen from synthesis gas?
Concept: undefined >> undefined
Match Column I with Column II for the given properties/applications mentioned therein.
| Column I | Column II |
| (i) \[\ce{H}\] | (a) Used in the name of perhydrol. |
| (ii) \[\ce{H2}\] | (b) Can be reduced to dihydrogen by \[\ce{NaH}\]. |
| (iii) \[\ce{H2O}\] | (c) Can be used in hydroformylation of olefin. |
| (iv) \[\ce{H2O2}\] | (d) Can be used in cutting and welding. |
Concept: undefined >> undefined
Atomic hydrogen combines with almost all elements but molecular hydrogen does not. Explain.
Concept: undefined >> undefined
