Please select a subject first
Advertisements
Advertisements
During the reaction of some metals with dilute hydrochloric acid, following observations are made.
Silver metal does not show any change
Explain this observation giving suitable reason.
Concept: undefined >> undefined
During the reaction of some metals with dilute hydrochloric acid, following observations are made.
The temperature of the reaction mixture rises when aluminium (Al) is added.
Explain this observation giving suitable reason.
Concept: undefined >> undefined
Advertisements
During the reaction of some metals with dilute hydrochloric acid, following observations are made.
The reaction of sodium metal is found to be highly explosive
Explain this observation giving suitable reason.
Concept: undefined >> undefined
During the reaction of some metals with dilute hydrochloric acid, following observations are made.
Some bubbles of a gas are seen when lead (Pb) is reacted with the acid.
Explain this observation giving suitable reason.
Concept: undefined >> undefined
Why do we store silver chloride in a dark coloured bottle?
Concept: undefined >> undefined
Zinc liberates hydrogen gas when reacted with dilute hydrochloric acid whereas copper does not. Explain why?
Concept: undefined >> undefined
A silver article generally turns black when kept in the open for a few days. The article when rubbed with toothpaste again starts shining.
- Why do silver articles turn black when kept in the open for a few days? Name the phenomenon involved.
- Name the black substance formed and give its chemical formula.
Concept: undefined >> undefined
What happens when zinc granules are treated with dilute solution of H2SO4, HCl, HNO3, NaCl and NaOH Also write the chemical equations if reaction oçcurs.
Concept: undefined >> undefined
On adding a drop of barium chloride solution to an aqueous solution of sodium sulphite, white precipitate is obtained :
- Write a balanced chemical equation for the reaction involved.
- What other name can be given to this precipitation reaction?
- On adding dilute hydrochloric acid to the reaction mixture, white precipitate disappears. Why?
Concept: undefined >> undefined
You are provided with two containers made up of copper and aluminium. You are also provided with solutions of dilute HCl, dilute HNO3, ZnCl2 and water. In which of the above containers, can these solutions be kept?
Concept: undefined >> undefined
For a current in a long straight solenoid N- and S-poles are created at the two ends. Among the following statements, the incorrect statement is
Concept: undefined >> undefined
What does the direction of thumb indicate in the right-hand thumb rule. In what way this rule is different from Fleming’s left-hand rule?
Concept: undefined >> undefined
Describe the activity that shows that a current-carrying conductor experiences a force perpendicular to its length and the external magnetic field. How does Fleming’s left-hand rule help us to find the direction of the force acting on the current carrying conductor?
Concept: undefined >> undefined
A simple motor is made in a school laboratory. A coil of wire is mounted on an axle between the poles of a horseshoe magnet, as illustrated.

In the example above, coil ABCD is horizontal and the battery is connected as shown.
- For this position, state the direction of the force on the arm AB.
- Why does the current in the arm BC not contribute to the turning force on the coil?
Concept: undefined >> undefined
In the redox reaction:
MnO2 + 4HCl → MnCl2 + 2H2O + Cl2
Concept: undefined >> undefined
A copper wire is held between the poles of a magnet
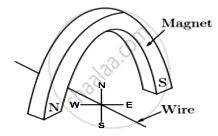
The current in the wire can be reversed. The pole of the magnet can also be changed over. In how many of the four directions shown can the force act on the wire?
Concept: undefined >> undefined
Which of the following pattern correctly describes the magnetic field around a long straight wire carrying current?
Concept: undefined >> undefined
State whether the given chemical reaction is a redox reaction or not. Justify your answer.
\[\ce{MnO_2 + 4HCl->MnCl_2 +2H_2O + Cl_2}\]
Concept: undefined >> undefined
A reddish brown metal used in electrical wires when powdered and heated strongly turns black. When hydrogen gas is passed over this black substance, it regains its original colour. Based on this information answer the following questions:
- Name the metal and the black substance formed.
- Write balanced chemical equations for the two reactions involved in the above information
Concept: undefined >> undefined
State the change in colour observed in following case mentioning the reason:
Copper powder is strongly heated in air.
Concept: undefined >> undefined
