Advertisements
Advertisements
Write a balanced equation for the action of dilute hydrochloric acid on sodium sulphide.
Concept: General Preparation of Hydrogen Chloride Gas
State your observation When moist starch iodide paper is introduced into chlorine gas.
Concept: General Preparation of Hydrogen Chloride Gas
Study the figure given below and answer the questions which follow:
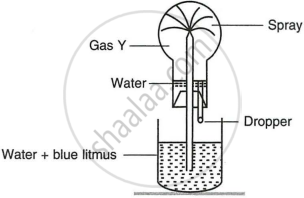
- Identify the gas Y.
- What property of gas Y does this experiment demonstrate?
- Name another gas which has the same property and can be demonstrated through this experiment.
Concept: Physical Properties of Hydrogen Chloride Gas
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
Concept: General Preparation of Hydrogen Chloride Gas
Dry hydrogen chloride gas can be collected by ________ displacement of air. (downward / upward)
Concept: General Preparation of Hydrogen Chloride Gas
For the preparation of hydrochloric acid in the laboratory:
What arrangement is done to dissolve hydrogen chloride gas in water?
Concept: General Preparation of Hydrogen Chloride Gas
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
Concept: General Preparation of Hydrogen Chloride Gas
When sodium chloride is heated with concentrated sulphuric acid below 200°C, one of the products formed is ______.
Concept: Chemical Properties of Hydrogen Chloride Gas
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Mangenese dioxide and copper (II) oxide. (using concentrated HCl)
Concept: Hydrogen Chloride
How will you distinguished between dilute HCl and dilute H2SO4 using lead nitrate solution?
Concept: Properties of Hydrochloric Acid > Physical Properties
State your observation of the following case:
When calcium hydroxide is heated with ammonium chloride crystals.
Concept: Chemical Properties of Hydrogen Chloride Gas
State the following:
The drying agent used in the laboratory preparation of HCl gas.
Concept: Physical Properties of Hydrogen Chloride Gas
Write a balanced equation for the following conversion:
Sodium chloride from sodium sulphite and dilute hydrochloric acid.
Concept: Properties of Hydrochloric Acid > Physical Properties
Identify the terms for the following:
A method used to collect HCl gas.
Concept: Physical Properties of Hydrogen Chloride Gas
Distinguish between the following:
Dilute HCl and dilute HNO3 [using silver nitrate solution]
Concept: Properties of Hydrochloric Acid > Physical Properties
Differentiate between the following pair based on the criteria given in the bracket:
Sulphuric acid and hydrochloric acid [type of salts formed]
Concept: Hydrochloric Acid
Convert the following reaction into a balanced chemical equation:
Ferrous sulphide to hydrogen sulphide using hydrochloric acid.
Concept: Properties of Hydrochloric Acid > Physical Properties
The following table is related to an industrial process of an acid.
| Name of the process | Reactant | Catalyst | Final product |
| (a) | SO2 + O2 | (b) | (c) |
Identify (a), (b) and (c).
Concept: Uses of Hydrochloric Acid
Assertion (A): Dry hydrogen chloride gas is collected by the upward displacement of air.
Reason (R): Hydrogen chloride gas is lighter than air.
Concept: Hydrochloric Acid
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is ______.
Concept: General Preparation of Hydrogen Chloride Gas
