Advertisements
Advertisements
प्रश्न
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Mangenese dioxide and copper (II) oxide. (using concentrated HCl)
Advertisements
उत्तर
MnO2 with conc. HCl gives Cl2 gas where as Cu2O does not.
APPEARS IN
संबंधित प्रश्न
Name a chloride which is solube in excess of ammonium hydroxide
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb3O4 + HCl ->}\]
Name the following :
Acid present in the stomach of mammals.
Name the following:
Acid used to extract glue from bones.
Give a balanced equation when dilute hydrochloric acid is added to : Calcium carbonate
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
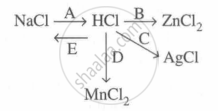
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.