Advertisements
Advertisements
Give a balanced chemical equation for Laboratory preparation of Nitric acid.
Concept: Laboratory Preparation of Nitric Acid
Write balanced chemical equations to show : The oxidizing action of conc. Sulphuric acid on carbon
Concept: Chemical Properties of Sulphuric Acid
Give one word or phrase for the following:
The amount of energy released when an atom in the gaseous state accepts an electron to form an anion.
Concept: Periodic Properties >> Electron Affinity
Identify the following:
The energy required to remove an electron from a neutral gaseous atom.
Concept: Periodic Properties >> Ionisation Potential (Ionisation Energy)
The electron affinity of an element X is greater than that of element Y.
How is the oxidising power of X likely to compare with that of Y?
Concept: Periodic Properties >> Electron Affinity
The electron affinity of an element X is greater than that of element Y.
How is the electronegativity of X likely to compare with that of Y?
Concept: Periodic Properties >> Electron Affinity
The electron affinity of an element X is greater than that of element Y.
State whether X is likely to be placed to the left or to the right of Y in the periodic table.
Concept: Periodic Properties >> Electron Affinity
Give a reason for Inert gases do not form ions.
Concept: Coordinate Bond
The covalent bond in which the electrons are shared equally between the combining atoms.
Concept: The Covalent Bond
Identify the following:
A bond formed between two atoms by sharing of a pair of electrons, with both electrons being provided by the same atom.
Concept: Coordinate Bond
The salt prepared by the method of direct combination is _______.
Concept: Salts
Identify the following:
A salt formed by the complete neutralization of an acid by a base.
Concept: Classification of Salts
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Copper sulphate
Concept: Salts
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Sodium zincate
Concept: Salts
State your observations when Barium chloride solution is mixed with Sodium Sulphate Solution.
Concept: Action of Alkalis on Metal Oxides
| Abhishek was given a salt ‘X’ which was white in colour for analysis. On strong heating, it produced a yellow residue, a colourless gas and also a reddish-brown gas. The solution of the salt ‘X’, when tested with an excess of ammonium hydroxide, produced a chalky white insoluble precipitate. |
- Name the coloured gas that evolved when Abhishek heated the salt strongly.
- Which cation was present in the sample given to Abhishek?
- Identify the salt given to Abhishek for analysis.
Concept: Colours of the Salts and Their Solutions
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
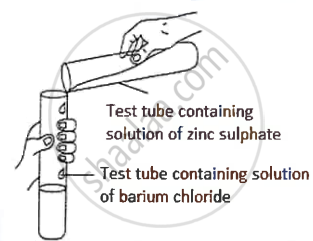
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
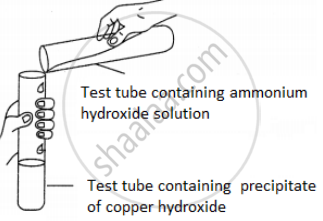
Concept: Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Concept: Vapour Density and Its Relation to Relative Molecular Mass
Name the main constituent metal in the following alloys:
Duralumin
Concept: Methods of Making Alloys
