Advertisements
Advertisements
प्रश्न
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is ______.
विकल्प
22:1
1:2.2
1:10
10:1
Advertisements
उत्तर
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is 1:10.
APPEARS IN
संबंधित प्रश्न
State one relevant observation for the following
When hydrogen sulfide gas is passed through lead acetate solution.
Write an equation for the reaction of hydrochloric acid on the silver nitrate solution.
Write an equation for the reaction of aqueous hydrochloric acid on lead nitrate solution.
Name the following:
Drying agent used to dry hydrogen chloride.
Name the following:
A chemical in which gold can be dissolved.
What is the function of HCI in preparation of aqua-regia?
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Magnesium sulphite
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
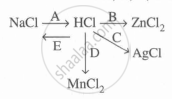
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.