Advertisements
Advertisements
Asha's mother read an article in the newspaper about a disaster that took place at Chernobyl. She could not understand much from the articles and asked a few questions from Asha regarding the article. Asha tried to answer her mother's questions based on what she learnt in Class XII Physics.
(a) What was the installation at Chernobyl where the disaster took place? What according to you, was the cause of this disaster?
(b) Explain the process of release of energy in the installation at Chernobyl.
(c) What according to you, were the values displayed by Asha and her mother?
Concept: Mass-energy and Nuclear Binding Energy >> Mass - Energy
Write the basic nuclear process underlying β+ and β– decays.
Concept: Radioactivity >> Beta Decay
Define the terms (i) half-life (T1/2) and (ii) average life (τ). Find out their relationships with the decay constant (λ).
Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
In a typical nuclear reaction, e.g.
`"_1^2H+"_1^2H ->"_2^3He + n + 3.27 \text { MeV },`
although number of nucleons is conserved, yet energy is released. How? Explain.
Concept: Mass-energy and Nuclear Binding Energy >> Mass - Energy
Write the relationship between the size of a nucleus and its mass number (A)?
Concept: Mass-energy and Nuclear Binding Energy >> Mass - Energy
In a given sample, two radioisotopes, A and B, are initially present in the ration of 1 : 4. The half lives of A and B are respectively 100 years and 50 years. Find the time after which the amounts of A and B become equal.
Concept: Law of Radioactive Decay
Distinguish between isotopes and isobars.
Concept: Atomic Masses and Composition of Nucleus
Two nuclei have different mass numbers A1 and A2. Are these nuclei necessarily the isotopes of the same element? Explain.
Concept: Atomic Masses and Composition of Nucleus
Explain the release of energy in nuclear fission and fusion on the basis of binding energy per nucleon curve.
Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
How mass defect is related to the stability of the nucleus?
Concept: Atomic Mass, Mass - Energy Relation and Mass Defect
With the help of a neat circuit diagram, explain the working of a photodiode.
Concept: Special Purpose P-n Junction Diodes
Meeta's father was driving her to school. At the traffic signal, she noticed that each traffic light was made of many tiny lights instead of a single bulb. When Meeta asked this question to her father, he explained the reason for this.
Answer the following questions based on above information:
(i) What were the values displayed by Meeta and her father?
(ii) What answer did Meeta's father give?
(iii) What are the tiny lights in traffic signals called and how do these operate?
Concept: Special Purpose P-n Junction Diodes
(i) Explain with the help of a diagram the formation of depletion region and barrier potential in a pn junction.
Concept: Semiconductor Diode
Why is zener diode fabricated by heavily doping both p- and n-sides of the junction?
Concept: Special Purpose P-n Junction Diodes
Draw the circuit diagram of zener diode as a voltage regulator and briefly explains its working ?
Concept: Zener Diode as a Voltage Regulator
How is a photodiode fabricated?
Concept: Special Purpose P-n Junction Diodes
Briefly explain its working. Draw its V - I characteristics for two different intensities of illumination ?
Concept: Special Purpose P-n Junction Diodes
In the circuit shown in the figure, identify the equivalent gate of the circuit and make its truth table.
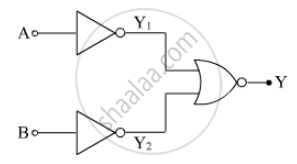
Concept: Integrated Circuits
Describe briefly with the help of a circuit diagram, how the flow of current carriers in a p-n-p transistor is regulated with emitter-base junction forward biased and base-collector junction reverse biased.
Concept: Junction Transistor >> Feedback Amplifier and Transistor Oscillator
