Advertisements
Advertisements
प्रश्न
Answer the following.
Match the following pairs:
| Name of polymer | Monomer |
| 1. Teflon | a. CH2 = CH2 |
| 2. PVC | b. CF2 = CF2 |
| 3. Polyester | c. CH2 = CHCl |
| 4. Polythene | d. C6H5OH and HCHO |
| 5. Bakelite | e. Dicarboxylic acid and polyhydoxyglycol |
Advertisements
उत्तर
| Name of polymer | Monomer |
| 1. Teflon | b. CF2 = CF2 |
| 2. PVC | c. CH2 = CHCl |
| 3. Polyester | e. Dicarboxylic acid and polyhydoxyglycol |
| 4. Polythene | a. CH2 = CH2 |
| 5. Bakelite | d. C6H5OH and HCHO |
APPEARS IN
संबंधित प्रश्न
Explain the following term: Homopolymers
Write the monomers of the following polymer :

Answer the following in one sentence.
Identify 'A' in the following reaction:
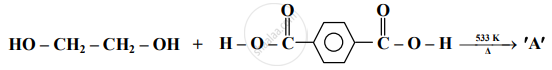
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write structure of natural rubber and neoprene rubber along with the name and structure of their monomers.
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Write the structure of isoprene and the polymer obtained from it.
Nylon 6, 6 is a condensation polymer of hexamethylenediamine and _____________
Monomer used for preparation of polyacrylonitrile is _____________
Write chemical reaction for preparation of the following.
Neoprene
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Explain vulcanization of rubber.
Define rubber.
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Which of the following polymer is used in paints?
The INCORRECT match for the polymer with its application is:
Identify the INCORRECT match.
Which of the following is the monomer of neoprene?
Which of the following polymers is obtained from chloroprene?
Novolac is obtained from ____________.
Which of the following compounds is used to prepare orlon?
Which among the following monomers is used to prepare Teflon?
Which of the following monomers is used in the manufacture of Neoprene rubber?
The commercial name of polyacrylonitrile is ______.
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Which of the following is not a copolymer?
Which of the following products is formed when benzaldehyde is treated with CH3MgBr and the addition product so obtained is subjected to acid hydrolysis?
Which of the following polymer has ester linkage?
Which of the following polymers is synthesized using a free radical polymerisation technique?
The catalyst used for the polymerisation of olefins is ______.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name the compound which reacts with formaldehyde to produce ethyl alcohol.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
Name and draw structure of the repeating unit in natural rubber.
The monomer used in preparation of teflon is ______.
Write the structure and name of the monomer of Nylon-6.
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Which from following statements is NOT true regarding neoprene?
