Advertisements
Advertisements
प्रश्न
How the following conversion can be carried out?
2-Chloropropane to 1-propanol
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[Alc{.} KOH, \Delta][Dehydrohalogenation] \underset{Propene}{CH3 - CH = CH2} ->[HBr, Peroxide] \underset{1-Bromopropane}{CH3 - CH2 - CH2 - Br} ->[KOH (aq.), \Delta][nucleophilic substitution] \underset{1-propanol}{CH3 - CH2 - CH2 - OH}}\\
|\phantom{...........................................................................................................................................................................................}\\
\ce{\underset{2-Chloropropane}{Cl}}\phantom{...........................................................................................................................................................................................}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
How do you convert:
2-bromobutane to but-2-ene
Write the main products when n-butyl chloride is treated with alcoholic KOH.
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
1-Bromo-1-methylcyclohexane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2-Chloro-2-methylbutane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2, 2, 3-Trimethyl-3-bromopentane
How the following conversion can be carried out?
1-Bromopropane to 2-bromopropane
Draw a neat, labelled energy profile diagram for SN1 reaction mechanism.
Observe the following compounds and answer the questions given below.

(I)
\[\ce{\underset{\text{(II)}}{CH3 - CH2 - Br}}\]
- Identify the type of halides.
- Explain the nature of the C – Br bond in both of these halides.
- Which of these compounds will undergo aqueous alkaline hydrolysis readily? Write the reaction in support of your answer.

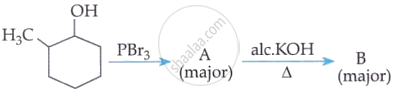
'A' is:
Identify the major product formed when 2-cyclohexylchloroethane undergoes a dehydrohalogenation reaction. Name the reagent which is used to carry out the reaction.
Elimination of bromine from 2-bromobutane results in the formation of ______.
A primary alkyl halide would prefer to undergo ______.
Reaction of trans-2-phenyl-1-bromocyclopentane with alcoholic KOH produces.
Which of the following alkyl halides will undergo SN1 reaction most readily?
An SN1 reaction at the asymmetric carbon of an enantiomerically pure chiral alkyl halide gives a product ______.
Major products A and B formed in the following reaction sequence are: