Advertisements
Advertisements
प्रश्न
For the complex [Fe(CN)6]3–, write the hybridization type, magnetic character and spin nature of the complex. (At. number : Fe = 26).
Advertisements
उत्तर
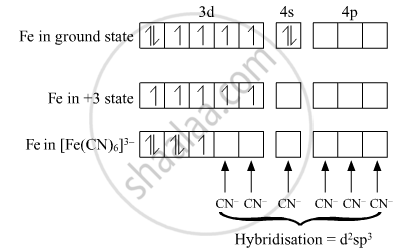
Hybridisation:- d2sp3
Magnetic character:- Paramagnetic
Spin nature of complex:- Low-spin complex
APPEARS IN
संबंधित प्रश्न
Write the hybridization and magnetic behaviour of the complex [Ni(CO)4].
(At.no. of Ni = 28)
Write the hybridization and shape of the following complexe : [Ni(CN)4]2–
(Atomic number : Co = 27, Ni = 28)
Explain [Co(NH3)6]3+ is an inner orbital complex, whereas [Ni(NH3)6]2+ is an outer orbital complex.
[At. No.: Co = 27, Ni = 28]
Why is [NiCl4]2− paramagnetic while [Ni(CN)4]2− is diamagnetic? (Atomic number of Ni = 28)
Write the hybridization and magnetic character of the following complexes:
[Fe(H2O)6]2+
(Atomic no. of Fe = 26)
Which of the following options are correct for \[\ce{[Fe(CN)6]^{3-}}\] complex?
(i) d2sp3 hybridisation
(ii) sp3d2 hybridisation
(iii) paramagnetic
(iv) diamagnetic
Magnetic moment of \[\ce{[MnCl4]^{2-}}\] is 5.92 BM. Explain giving reason.
Explain why \[\ce{[Fe(H2O)6]^{3+}}\] has magnetic moment value of 5.92 BM whereas \[\ce{[Fe(CN)6]^{3-}}\] – has a value of only 1.74 BM.
The correct order of magnetic moment (spin only value in B.m.) is:
Given below are two statements:
Statement I: Ferromagnetism is considered as an extreme form of paramagnetism.
Statement II: The number of unpaired electrons in a Cr2+ ion (Z = 24) is the same as that of a Nd3+ ion (Z = 60).
In the light of the above statements, choose the correct answer from the options given below:
