Advertisements
Advertisements
प्रश्न
Explain the following with an example.
Kolbe’s reaction.
Advertisements
उत्तर
Phenoxide ion generated by treating phenol with sodium hydroxide is even more reactive than phenol towards electrophilic aromatic substitution. Hence, it undergoes electrophilic substitution with carbon dioxide, a weak electrophile. Ortho hydroxybenzoic acid is formed as the main reaction product.

संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
Write the structures of A, B, C, D and E in the following reactions:

Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Write the equation involved in the following reaction:
Kolbe’s reaction
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:
When Phenol is distilled with zinc dust, it gives:


Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 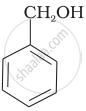 |
| (C) |  |
| (D) |  |
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why?
Convert the following:
Phenol to N-phenylethanamide.
Attacking species in nitration of benzene in presence of fuming HNO3 is
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
Why ortho-nitrophenol is steam volatile while para-nitrophenol is not?
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
