Advertisements
Advertisements
प्रश्न
Draw the geometrical isomers of [Co(en)2Cl2]2+. Which geometrical isomer of [Co(en)2Cl2]2+ is not optically active and why?
Advertisements
उत्तर
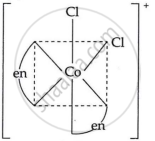 |
 |
| cis-form | trans-form |
The trans isomer of [Co(en)2Cl2]2+ is not optically active because of the plane of symmetry.
APPEARS IN
संबंधित प्रश्न
Explain cationic complexes and anionic complexes of co-ordination compounds.
When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3 moles of AgCl are precipitated per mole of the compound. Write the structural formula of the coordination compound.
Draw the geometrical isomers of complex \[\ce{[Co(en)2Cl2]+}\].
Define the following term:
Anomers
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [CoCl2(en)2]+.
Why is Butan-1-ol optically inactive but Butan-2-ol is optically active?
The IUPAC name for [CoCl(NO2)(en)2]Cl is ____________.
Assertion: Addition of bromine water to 1-butene gives two optical isomers.
Reason: The product formed contains two asymmetric carbon atoms.
