Advertisements
Advertisements
प्रश्न
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Phosphorus – \[\ce{^31_15P}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Advertisements
उत्तर
Phosphorus – \[\ce{^31_15P}\]
Atomic number Z = p = e = 15
Mass number A = p + n
31 = 15 + n
∴ n = 31 − 15 = 16
P
15 = 2, 8, 5
(K, L, M)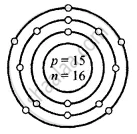
APPEARS IN
संबंधित प्रश्न
Fill in the blanks.
Isotopes are the atoms of ______ element having the ______ atomic number but ______ mass number.
Multiple Choice Questions
The sum of number of protons and number of neutrons present in the nucleus of an atom is called its
Name or state the following:
Elements having same mass number but different atomic number
If an atom ‘A’ has mass number 35 and atomic number 17, state the number of protons, electrons and neutrons it contains.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
What is atomic number?
The atomic number of an element is 9, it has 10 neutrons. Find the element from the periodic table. What will be its mass number?
Every atom has an equal number of protons and electrons. The nature of the atom is ______.
State the term for the following:
The amount of substance which contains the same number of units as the number of atoms in carbon-12.
Element ‘P' has electronic configuration 2,8,8,1. The number of chlorine atoms present in the chloride of ‘P’ is ______.
