Advertisements
Advertisements
प्रश्न
Draw isomers of the following.
\[\ce{Pt(NH3)2ClNO2}\]
Advertisements
उत्तर
Cis and trans isomers of \[\ce{Pt(NH3)2ClNO2}\]
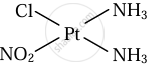 |
 |
| Cis isomer | Trans isomer |
APPEARS IN
संबंधित प्रश्न
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Define the term Hydrated isomers.
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Draw optical isomers of [Co(en)3]3+.
Which one of the following will give a pair of enantiomorphs?
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
What are hydrate isomers? Explain with an example.
Which would exhibit coordination isomerism?
The compound(s) that exhibit(s) geometrical isomerism is (are):
(I) [Pt(en)Cl2]
(II) [Pt(en)2]Cl2
(III) [Pt(en)2Cl2]
(IV) [Pt(NH3)2Cl2]
Which of the following is NOT a pair of structural isomers?
How many isomers are possible for an alkane having molecular formula C5H12?
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
____________ isomers are formed when the ligand has two different donor atoms.
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
The correct structure of Fe(CO)5 is
Which compound would exhibit optical isomers?
The one that is not expected to show isomerism is ______.
Define Distereoisomers.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
What are structural isomers?
Draw the structure of cis isomers of Pt(NH3)2Cl2.
Draw the structure of trans isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Which one of the following complex ions has geometrical isomers?
