Advertisements
Advertisements
प्रश्न
Classify the following as primary, secondary and tertiary alcohol:
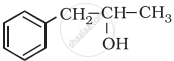
विकल्प
Primary alcohol
Secondary alcohol
Tertiary alcohol
Advertisements
उत्तर
Secondary alcohol
APPEARS IN
संबंधित प्रश्न
Classify the following as primary, secondary and tertiary alcohol:
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{....}\\
\ce{CH3}\phantom{.}
\end{array}\]
Identify allylic alcohols in the following examples.
Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
The alcohol used in thermometers is _______.
(A) methanol
(B) ethanol
(C) propanol
(D) butanol
How many isomers of C5H11OH will be primary alcohols?
How many alcohol(s) with molecular formula \[\ce{C4H10O}\] is chiral in nature?
Number of metamers represented by molecular formula C4H10O is ____________.
Lower molecular mass alcohols are ______.
How much ethyl alcohol must be added to 1 litre of water so that the solution will freeze at -14°C? (Kf for water = 1.86°C/mol)
Arrange the following in the increasing order of their property indicated:
Benzoic acid, Phenol, Picric acid, Salicylic acid (pka values).
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
Glycerol is not used in which of the following cares
Main constituent of dynamite is.
Washing soap can be prepared by saponification with alkali of the oil.
Write IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
