Advertisements
Advertisements
प्रश्न
How many alcohol(s) with molecular formula \[\ce{C4H10O}\] is chiral in nature?
विकल्प
1
2
3
4
Advertisements
उत्तर
1
Explanation:
\[\begin{array}{cc}
\ce{CH3-\overset{∗}{C}H-CH2-CH3}\\
|\phantom{........}\\
\ce{OH}\phantom{......}
\end{array}\]
has chiral centre (one).
APPEARS IN
संबंधित प्रश्न
Which of the followings is a trihydric alcohol ?
(a) n-propyl alcohol
(b) Glycerol
(c) Glycol
(d) Glycine
The alcohol used in thermometers is _______.
(A) methanol
(B) ethanol
(C) propanol
(D) butanol
Write a chemical equation for the action of neutral ferric chloride on phenol.
Which of the following is dihydric alcohol?
Lower molecular mass alcohols are ______.
How much ethyl alcohol must be added to 1 litre of water so that the solution will freeze at -14°C? (Kf for water = 1.86°C/mol)
Arrange the following in the increasing order of their property indicated:
Benzoic acid, Phenol, Picric acid, Salicylic acid (pka values).
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
Distinction b/w primary, secondary and tertiary alcohol is done by:-
Main constituent of dynamite is.
Which is used an antifreeze
Washing soap can be prepared by saponification with alkali of the oil.
Write IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3 \phantom{..}OH \phantom{..}CH3}
\end{array}\]
Classify the following as primary, secondary and tertiary alcohol:
CH3 – CH2 – CH2 – OH
Classify the following as primary, secondary and tertiary alcohol:
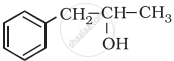
Classify the following as primary, secondary and tertiary alcohol:

