Advertisements
Advertisements
प्रश्न
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Advertisements
उत्तर
It is given that the compound (with molecular formula C9H10O) forms 2, 4-DNP derivative and reduces Tollen’s reagent. Therefore, the given compound must be an aldehyde.
Again, the compound undergoes Cannizzaro reaction and on oxidation gives 1, 2-benzenedicarboxylic acid. Therefore, the −CHO group is directly attached to a benzene ring and this benzaldehyde is ortho-substituted. Hence, the compound is 2-ethylbenzaldehyde.

2-Ethylbenzaldehyde
The given reactions can be explained by the following equations:

APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
Describe the following:
Cannizzaro reaction
Write the reactions involved in the following reactions: Clemmensen reduction
Complete the following reactions:

Write the chemical equations to illustrate the following name reactions:
Aldol condensation
Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
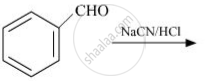
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Complete the following reaction:
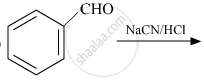
The products obtained in the Cannizzaro reaction are
The key step in cannizzaro reaction in the inter molecular shift qf
Which of the following does not give Cannizzaro reaction?
Write the chemical reaction involved in Cannizzaro reaction of methanal.
Calcium acetate, on heating, gives:
Benzaldehyde can be prepared by the hydrolysis of:
Which product is formed when the compound benzaldehyde is treated with concentrated aqueous KOH solution?
