Advertisements
Advertisements
प्रश्न
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Advertisements
उत्तर
Carboxylic acid having a-hydrogen are halogenated at a-position on treatment with 2 Br in presence of red phosphorous to give a -halocarboxylic acids. This is the HeII-Volhard-Zelinsky reaction.
\[\begin{array}{cc}
\phantom{.................}\ce{CH3}
\phantom{...........................}\ce{CH3}\\
\phantom{...............}|
\phantom{..............................}| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]CH3-C-COOH}\\
\phantom{.............................................} | \\
\phantom{..............................................} \ce{Br}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
Write the product(s) in the following reactions

Write the chemical equations to illustrate the following name reactions:
Aldol condensation
complete the following reaction:
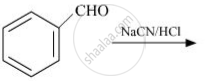
The key step in cannizzaro reaction in the inter molecular shift qf
Explain the following reaction:
Cannizzaro reaction
What happens when methanal undergoes cannizzaro reaction?
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
Which of the following does not give Cannizzaro reaction?
